Hypertension is a prevalent and common chronic disease that severely endangers human life and health. Globally, approximately 3.5 billion adults present suboptimal blood pressure levels, with a notable trend of younger onset [1]. The blood pressure regulatory system comprises the autonomic nervous system, vascular tone regulation system, renin-angiotensin-aldosterone system (RAAS), renal system and other functional modules. Among these, the RAAS exerts a pivotal role in arterial blood pressure regulation and extracellular fluid homeostasis maintenance [2].
Angiotensin-converting enzyme (ACE) serves as a core component of the RAAS. Inhibition of ACE activity reduces the biosynthesis of angiotensin II (Ang II), thereby inducing vasodilation and hypotensive effects. Currently, angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers constitute the predominant first-line antihypertensive pharmacotherapies [3-4]. Although these clinical agents exhibit satisfactory tolerability, nearly 20% of patients discontinue ACEI treatment due to adverse drug reactions such as chronic cough [5]. Accordingly, it is imperative to explore safer and more efficient hypotensive strategies and develop novel ACEIs characterized by superior efficacy, minimal side effects and high biosafety.
Accumulated studies have demonstrated that bioactive peptides liberated from food proteins possess multiple physiological functions, including antioxidant, antihypertensive, antibacterial, antithrombotic and immunomodulatory activities [6-7]. Angiotensin-converting enzyme inhibitory peptides (ACEIPs) are distinguished by high biological activity, favorable biosafety and efficient in vivo metabolic characteristics, rendering them increasingly prominent in the research and development of functional foods [8]. ACEIPs and their extracts can be efficiently prepared from dietary raw materials such as milk, meat and rice via enzymatic hydrolysis and microbial fermentation. These bioactive compounds exhibit potent ACE inhibitory capacity and prominent hypotensive properties, demonstrating broad application prospects in the food industry [9-11]. Nevertheless, the production of animal-derived proteins entails excessive energy consumption and substantial greenhouse gas emissions, which conflict with contemporary environmental sustainability concepts [12]. Plant-based raw materials, particularly cereal crops represented by rice, have garnered escalating research attention for ACEIP preparation by virtue of abundant raw material reserves and eco-friendly production advantages [7,13-14]. This review systematically summarizes the regulatory mechanisms of the blood pressure homeostasis system, the hypotensive pathways of ACEIPs, as well as their structural and functional characteristics. Particular emphasis is placed on elaborating the research advances in the preparation, separation and purification of rice-derived ACEIPs through enzymatic hydrolysis and fermentation technologies. This work aims to provide theoretical foundations for the industrial development of food-originated ACEIP products and the high-value extension of the rice industrial chain.
一.Blood Pressure Regulation and Hypotensive Mechanisms
1.Regulatory Mechanisms of the Blood Pressure System
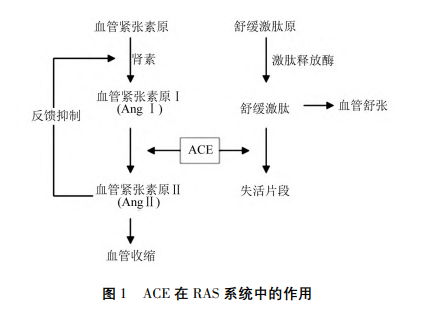
The inhibitory efficacy against ACE is primarily associated with two pivotal blood pressure regulatory axes: the renin-angiotensin system (RAS) and nitric oxide system (NOS). As illustrated in Figure 1, renin, a proteolytic enzyme synthesized by juxtaglomerular cells, catalyzes the conversion of angiotensinogen into angiotensin I (Ang I). ACE further mediates the transformation of Ang I into Ang II, which subsequently activates angiotensin II type 1 receptors (AT1R) and triggers a cascade of pathological responses including vasoconstriction, tissue fibrosis, inflammatory infiltration and reactive oxygen species (ROS) overproduction. Meanwhile, moderate levels of Ang II bind to angiotensin II type 2 receptors (AT2R) to counterbalance AT1R-mediated biological effects, thereby sustaining the physiological homeostasis of the RAS [15].
Zinc metalloprotease ACE2 is capable of degrading Ang II to generate Ang-(1-7), or directly converting Ang I into Ang-(1-9). The intermediate product Ang-(1-9) can be further hydrolyzed by ACE to produce Ang-(1-7). Functionally opposite to Ang II, Ang-(1-7) specifically couples with corresponding G-protein-coupled receptors to exert vasodilatory, anti-inflammatory and anti-fibrotic bioactivities, and is ultimately degraded by ACE into inactive Ang-(1-5) fragments [16]. Hence, ACE occupies a more dominant regulatory position than ACE2 within the RAS cascade. Both aminopeptidase A (AP-A) and aminopeptidase N (AP-N) catalyze the biotransformation of Ang II into Ang IV. Additionally, Ang IV binds to insulin-regulated aminopeptidase receptors (IRAP), resulting in adverse physiological outcomes such as vasoconstriction, inflammatory responses and ROS accumulation [17].
2.Structural Characteristics of ACEIPs
The physiological functions of ACEIPs are intrinsically correlated with their polypeptide structures, with key determinants including molecular weight, amino acid composition and sequence arrangement. The bioactivity of ACE inhibitory peptides is closely related to molecular mass. Multiple studies have verified that short-chain peptides containing 2 to 12 amino acid residues (with molecular weights generally below 3000 Da) can optimally bind to the active sites of ACE [18-19]. In most ACEIP sequences, the last three amino acid residues at the C-terminus are hydrophobic amino acids, which bind to the terminal catalytic sites of ACE to achieve targeted enzyme inhibition.
ACEIPs with proline (Pro) at the C-terminus have been validated to possess enhanced inhibitory potency and superior resistance to digestive enzyme degradation [20-21]. Hydroxyproline (Hyp) plays a critical role in the molecular interaction with the ACE active pocket, and its presence at the penultimate position of the C-terminus significantly enhances ACE inhibitory activity [22-23]. For tetrapeptide ACEIPs, specific residue combinations confer potent inhibitory effects: tyrosine (Tyr) or cysteine (Cys) at the first position, histidine (His), tryptophan (Trp) or methionine (Met) at the second position, isoleucine (Ile), leucine (Leu), valine (Val) or methionine (Met) at the third position, and tryptophan (Trp) at the fourth position [24-25]. Molecular docking analysis further clarifies the binding modes of ACEIPs. As depicted in Figure 2, the tripeptide Tyr-Ser-Lys (YSK) exerts ACE inhibitory effects mainly via hydrogen bond formation within the ACE active pocket [26].
Current research also indicates that hydrophobic amino acids at the N-terminus endow polypeptides with strong ACE inhibitory capacity, while basic amino acid residues at the N-terminus can strengthen molecular affinity to ACE and further elevate antihypertensive bioactivity [18-19,27]. Beyond the 20 canonical amino acids, non-canonical amino acids also modulate ACEIP bioactivity. For instance, citrulline (Cit) with neutral net charge increases protein hydrophobicity upon accumulation; ornithine (Orn), a typical basic amino acid, can enhance ACEIP activity when located at the N-terminus. Both non-canonical amino acids exhibit antioxidant and vascular-protective properties, which may assist in alleviating adverse physiological side effects [28-30].
3.Preparation Methods of Rice-Derived ACEIPs
Major fabrication technologies for bioactive peptides include solvent extraction, enzymatic hydrolysis, microbial fermentation, chemical synthesis and genetic engineering recombination [31]. Solvent extraction is characterized by simple principles and convenient operational procedures; in vitro synthetic approaches such as chemical synthesis and recombinant genetic engineering offer distinct advantages of short production cycles and high yield, effectively compensating for the limitations of natural peptide acquisition [32]. In contrast, enzymatic hydrolysis and microbial fermentation have evolved as mature industrial preparation methods due to high biosafety, robust biological activity and specific catalytic specificity. With the rapid advancement of biotechnology, innovative strategies for natural peptide production continue to emerge. At present, enzymatic hydrolysis and microbial fermentation represent the mainstream technical routes for ACEIP production using food-derived raw materials.
二. Preparation of Rice-Derived ACEIPs via Enzymatic Hydrolysis
1. Utilization of Different Rice Fractions
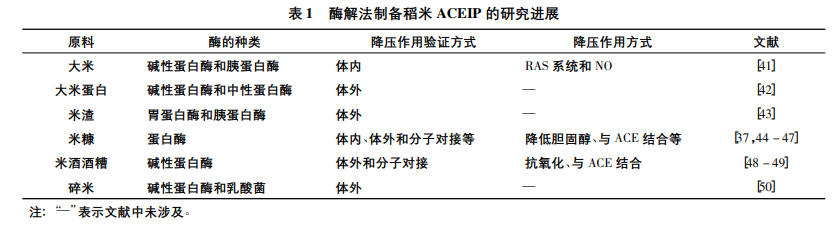
Rice contains a relatively low protein content (approximately 8%), whereas the nutritional value and health-promoting properties of rice proteins have been widely recognized [33]. Native rice proteins exhibit poor solubility and digestibility under neutral conditions, and enzymatic modification can effectively improve their functional and biological properties. Proteins isolated from rice can be hydrolyzed into abundant bioactive peptides, which exert dose-dependent ACE inhibitory effects at appropriate concentrations [34-35]. As summarized in Table 1, rice protein hydrolysates prepared via hydrolysis with multiple commercial enzyme preparations display efficient in vivo and in vitro hypotensive activities. Several typical rice-derived ACEIPs have been successfully identified, including Ile-His-Arg-Phe (IHRF), Val-Asn-Pro (VNP) and Val-Trp-Pro (VWP) [36-37].
Preliminary separation of rice protein hydrolysates can be achieved through ultrafiltration and DA201 macroporous adsorption resin, followed by two-step reversed-phase high-performance liquid chromatography (RP-HPLC) purification to obtain two high-purity tripeptides with prominent ACE inhibitory activity [37]. Six novel ACE inhibitory peptides have been separately isolated from red yeast rice and traditional Korean rice wine in previous studies [38-39]. Moreover, oral administration of fermented brown rice aqueous extracts and rice residue protein hydrolysates containing peptide components yields marked hypotensive outcomes in spontaneously hypertensive rats (SHRs) [40].
Proteins extracted from rice processing by-products present favorable biological characteristics (Table 1). Rice bran, rice residue and distillers’ grains are cost-effective and sustainable raw material sources for ACEIP production. Unhydrolyzed rice bran proteins demonstrate weaker ACE inhibitory activity compared with their enzymatic hydrolysates. Commercial protease-treated rice bran protein hydrolysates feature low relative molecular weight and potent in vitro ACE inhibitory activity. Enzymatically modified rice bran proteins can be developed as high-value functional food ingredients with superior bioavailability and absorption efficiency [51].
Novel bioactive peptides are continuously discovered from rice by-products. For example, a novel ACE inhibitory peptide with the sequences of Ile-Thr-Leu (ITL) or Leu-Thr-Ile (LTI) was purified from alkaline protease-hydrolyzed rice bran protein via size-exclusion chromatography and RP-HPLC, whose in vitro ACE inhibitory potency was comparable to that of enalapril maleate [47,52]. Multiple dipeptides and tripeptides with validated in vivo and in vitro ACE inhibitory effects have been identified in rice bran enzymatic hydrolysates [53-54]. Rice bran protein hydrolysates exhibit efficient ACE suppression capacity, with pharmacological effects analogous to the classic hypotensive drug captopril [55]. Additionally, protein hydrolysates derived from brown rice, refined rice protein and germinated brown rice also possess inherent ACE inhibitory potential [56-59].
2.Application of Enzymatic Preparations
As listed in Table 1, proteases including alkaline protease, trypsin, neutral protease and pepsin display unique cleavage sites and optimal reaction conditions, contributing to differential hypotensive performance in ACEIP preparation. Alkaline protease is the most widely adopted and high-efficiency protease in current research, which is closely associated with the poor solubility of rice proteins under acidic and neutral environments. A comparative study on the enzymatic hydrolysis efficiency of rice bran protein concentrate and soybean protein was conducted using alkaline protease, papain and commercial compound enzymes containing trypsin, thymoprotease and aminopeptidase. The results confirmed that alkaline protease possesses superior hydrolytic capacity and ACE inhibitory modification efficiency [60].
Enzymatically treated rice bran serves as an excellent natural source of bioactive peptides, showing promising therapeutic potential for hypertension and related metabolic disorders. Beyond ACE inhibitory activity, rice protein hydrolysates obtained via enzymatic degradation exhibit strong free radical scavenging capacity and reducing power. These multifunctional hydrolysates can be applied as functional food additives in nutritional foods and beverages, delivering comprehensive health benefits and satisfactory sensory quality [56-59].
三. Separation and Identification Technologies for ACEIPs
The conventional technical workflow for ACEIP separation and identification consists of three core steps: separation of rice-derived crude polypeptides using membrane separation technology, gel filtration chromatography and RP-HPLC; amino acid sequence identification via mass spectrometry; and determination of interaction sites and binding patterns through molecular docking simulation, which collectively assist the targeted separation and structural characterization of ACEIPs [26,37,47].
Innovative monitoring technologies have also been integrated into the enzymatic hydrolysis process. For instance, in-situ near-infrared spectroscopy combined with chemometric algorithms enables real-time detection of ACE inhibitory activity during dual-enzyme hydrolysis of rice-derived substrates. This integrated monitoring system can precisely regulate the hydrolysis termination point and adaptive transition parameters, thereby improving overall enzymatic hydrolysis efficiency [42]. Surface plasmon resonance and other biophysical technologies can simulate enzymatic hydrolysis and fermentation conditions to screen potential bioactive peptides, facilitate structural characterization and mechanism elucidation, and provide theoretical guidance for the functional prediction of ACE inhibitory peptides.
In silico screening strategies are increasingly applied in ACEIP research. By comparing acquired peptide sequences with published polypeptide databases from plant, animal and other food sources, bioinformatics databases such as BIOPEP and computer-aided matching tools can rapidly screen candidate bioactive peptides, whose actual biological activity is further verified through in vitro experiments to explore novel ACE inhibitory components [82-83]. Furthermore, systematic assessment and predictive analysis of the physicochemical properties, primary structures, toxicological risks and allergenicity of most ACE inhibitory peptides can be performed, so as to comprehensively guarantee the safety of final functional food products [84]. The combined application of these advanced separation and identification technologies greatly promotes the basic research and industrial development of rice-derived hypotensive functional foods and other bioactive food products.
四. Research Prospects and Limitations of Rice-Derived ACEIPs
The production of ACEIPs from rice and its processing by-products via enzymatic hydrolysis, microbial fermentation or combined biotechnological strategies presents enormous development potential. Consumers have gradually recognized the unique functional advantages and nutritional value of rice-derived functional products. Nevertheless, compared with animal-derived and other plant-derived bioactive peptides, research on rice-based ACEIPs prepared by enzymatic hydrolysis and fermentation remains relatively limited, leaving substantial room for in-depth exploration and industrial application.
The structure-activity relationship of ACEIPs and their functional regulatory mechanisms require further systematic investigation. In particular, in vivo metabolic pathways and clinical evidence of fermented rice-derived ACEIPs are still insufficient. Future research on the bioactivity of ACEIPs is expected to rely on multi-omics technologies including genomics and metabolomics to clarify their biosynthesis mechanisms and structural contributions to physiological functions. Transcriptomics and other molecular biological approaches, combined with computational simulation software, can be utilized to analyze the interactive relationships among fermentative microorganisms, optimize the targeted biosynthesis of rice-derived bioactive peptides, and elucidate their intracellular metabolism and transmembrane transport mechanisms.
Further in-depth research is urgently required to enhance peptide production efficiency and oral bioavailability, evaluate the in vivo bioaccessibility and long-term hypotensive efficacy of rice-derived ACEIPs. Collectively, ongoing mechanistic and applied research will provide solid theoretical and technical support for the innovative development and clinical auxiliary application of health care products and adjuvant antihypertensive agents based on rice bioactive peptides.
References:
[1] DZAU V J, HODGKINSON C P. Precision hypertension[J]. Hypertension, 2024, 81(4): 702-708.
[2] OLFAT A, MOSTAGHIM T, SHAHRIARI S, et al. Extraction of bioactive compounds of Hypnea flagelliformis by ultrasound-assisted extraction coupled with natural deep eutectic solvent and enzyme inhibitory activity[J]. Algal Research, 2024, 78: 103388.
[3] OPARIL S, ACELAJADO M C, BAKRIS G L, et al. Hypertension[J]. Nature Reviews Disease Primers, 2018, 4: 18014.
[4] WANG J W, YE Y Y, CHEN X F, et al. Diverse trends in antihypertensive medication usage among U.S. adults with hypertension by socioeconomic status and comorbidities, 1999—2020[J]. Blood Pressure, 2025, 34(1): 2506081.
[5] FABIAN I M, MADDOX K, ROBICHEAUX C, et al. Stevens-Johnson syndrome from combined allopurinol and angiotensin-converting enzyme inhibitors: A narrative review[J]. Cureus, 2024, 16(1): e51899.
[6] VILLANUEVA A, RIVERO-PINO F, MARTIN M E, et al. Identification of the bioavailable peptidome of chia protein hydrolysate and the in silico evaluation of its antioxidant and ACE inhibitory potential[J]. Journal of Agricultural and Food Chemistry, 2024, 72(6): 3189-3199.
[7] RAMAKRISHNAN V V, HOSSAIN A, DAVE D, et al. Salmon processing discards: a potential source of bioactive peptides-a review[J]. Food Production, Processing and Nutrition, 2024, 6(1): 22.
[8] XIANG L, QIU Z C, ZHAO R J, et al. Advancement and prospects of production, transport, functional activity and structure-activity relationship of food-derived angiotensin converting enzyme (ACE) inhibitory peptides[J]. Critical Reviews in Food Science and Nutrition, 2023, 63(10): 1437-1463.
[9] HERES A, YOKOYAMA I, GALLEGO M, et al. Antihypertensive potential of sweet Ala-Ala dipeptide and its quantitation in dry-cured ham at different processing conditions[J]. Journal of Functional Foods, 2021, 87: 104818.
[10] SHANMUGAM V P, KAPILA S, KEMGANG T S, et al. Isolation and characterization of angiotensin converting enzyme inhibitory peptide from buffalo casein[J]. International Journal of Peptide Research and Therapeutics, 2021, 27(2): 1481-1491.
[11] KAISER S, MARTIN M, LUNOW D, et al. Tryptophan-containing dipeptides are bioavailable and inhibit plasma human angiotensin-converting enzyme in vivo[J]. International Dairy Journal, 2016, 52: 107-114.
[12] ZHANG D J, JIANG K, LUO H, et al. Replacing animal proteins with plant proteins: Is this a way to improve quality and functional properties of hybrid cheeses and cheese analogs?[J]. Comprehensive Reviews in Food Science and Food Safety, 2024, 23(1): e13262.
[13] PREKUMAR J, MALINI M, JOSHY A. A critical review on food protein-derived antihypertensive peptides[J]. Drug Invention Today, 2019, 12(3): 474-479.
[14] CHUKWUMA C I. Antioxidative, metabolic and vascular medicinal potentials of natural products in the non-edible wastes of fruits belonging to the Citrus and Prunus genera: A review[J]. Plants, 2024, 13(2): 191.
[15] FARIA-COSTA G, LEITE-MOREIRA A, HENRIQUES-COELHO T. Efeitos cardiovasculares do receptor tipo 2 da angiotensina[J]. Revista Portuguesa de Cardiologia, 2014, 33(7/8): 439-449.
[16] METLOCK F E, HINNEH T, BENJASIRISAN C, et al. Impact of social determinants of health on hypertension outcomes: A systematic review[J]. Hypertension, 2024, 81(8): 1675-1700.
[17] HALLBERG M, LARHED M. From angiotensin IV to small peptidemimetics inhibiting insulin-regulated aminopeptidase[J]. Frontiers in Pharmacology, 2020, 11: 590855.
[18] SUN S Q, XU X T, SUN X, et al. Preparation and identification of ACE inhibitory peptides from the marine macroalga Ulva intestinalis[J]. Marine Drugs, 2019, 17(3): 179.
[19] TOOPCHAM T, MES J J, WICHERS H J, et al. Bioavailability of angiotensin I-converting enzyme (ACE) inhibitory peptides derived from Virgibacillus halodenitrificans SK1-3-7 proteinases hydrolyzed tilapia muscle proteins[J]. Food Chemistry, 2017, 220: 190-197.
[20] MIRALLES B, AMIGO L, RECIO I. Critical review and perspectives on food-derived antihypertensive peptides[J]. Journal of Agricultural and Food Chemistry, 2018, 66(36): 9384-9390.
[21] JAO C L, HUANG S L, HSU K C. Angiotensin I-converting enzyme inhibitory peptides: Inhibition mode, bioavailability, and antihypertensive effects[J]. BioMedicine, 2012, 2(4): 130-136.
[22] CIAU-SOLÍS N, RODRÍGUEZ-CANTO W, FERNÁNDEZ-MARTÍNEZ L, et al. Inhibitory activity of angiotensin-I converting enzyme (ACE-I) from partially purified Phaseolus lunatus peptide fractions[J]. Process Biochemistry, 2024, 139: 44-50.
[23] TAGA Y, HAYASHIDA O, ASHOUR A, et al. Characterization of angiotensin-converting enzyme inhibitory activity of X-hyp-gly-type tripeptides: Importance of collagen-specific prolyl hydroxylation[J]. Journal of Agricultural and Food Chemistry, 2018, 66(33): 8737-8743.
[24] LIU C, ZHANG Z J, SHANG Y T, et al. Progress in the preparation, identification and biological activity of walnut peptides[J]. Journal of Future Foods, 2024, 4(3): 205-220.
[25] XIANG L, ZHENG Z J, GUO X J, et al. Two novel angiotensin I-converting enzyme inhibitory peptides from garlic protein: In silico screening, stability, antihypertensive effects in vivo and underlying mechanisms[J]. Food Chemistry, 2024, 435: 137537.
[26] WANG X M, CHEN H X, FU X G, et al. A novel antioxidant and ACE inhibitory peptide from rice bran protein: Biochemical characterization and molecular docking study[J]. LWT-Food Science and Technology, 2017, 75: 93-99.
[27] LEE S Y, HUR S J. Antihypertensive peptides from animal products, marine organisms, and plants[J]. Food Chemistry, 2017, 228: 506-517.
[28] Chen S R, Wei T, Li Q, et al. Process optimization and dynamic analysis of ACE inhibitory peptides produced by fermenting buffalo milk with Lactobacillus plantarum Strain B[J]. Cereals & Oils, 2024, 37(12): 89-96.
[29] LI H J, WANG Y S, WANG Y N, et al. Mechanical study of alisol B23-acetate on methionine and choline deficient diet-induced nonalcoholic steatohepatitis based on untargeted metabolomics[J]. Biomedical Chromatography, 2024, 38(1): e5763.
[30] BUTTERWORTH R F, CANBAY A. Hepatoprotection by L-ornithine L-aspartate in non-alcoholic fatty liver disease[J]. Digestive Diseases, 2019, 37(1): 63-68.
[31] MONDAL S, DAS S, NANDI A K. A review on recent advances in polymer and peptide hydrogels[J]. Soft Matter, 2020, 16(6): 1404-1454.
[32] FENG L, WANG Y, YANG J, et al. Overview of the preparation method, structure and function, and application of natural peptides and polypeptides[J]. Biomedecine & Pharmacotherapie, 2022, 153: 113493.
[33] TYAGI A, CHELLIAH R, BANAN-MWINE DALIRI E, et al. Antioxidant activities of novel peptides from Limosilactobacillus reuteri fermented brown rice: A combined in vitro and in silico study[J]. Food Chemistry, 2023, 404(Pt B): 134747.
[34] DALIRI E B, LEE B H, OH D H. Current trends and perspectives of bioactive peptides[J]. Critical Reviews in Food Science and Nutrition, 2018, 58(13): 2273-2284.
[35] KONTANI N, OMAE R, KAGEBAYASHI T, et al. Characterization of Ile-His-Arg-Phe, a novel rice-derived vasorelaxing peptide with hypotensive and anorexigenic activities[J]. Molecular Nutrition & Food Research, 2014, 58(2): 359-364.
[36] KUMAR A, SINGH N, JOSHI R. Evaluation of antioxidant and ACE inhibitory peptides in rice bran protein hydrolysate using non-targeted UHPLC-QTOF-IMS profiling, MALDI-TOF-TOF-MS/MS, and molecular docking study[J]. International Journal of Food Science & Technology, 2024, 59(6): 4300-4314.
[37] KUBA M, TANAKA K, SESOKO M, et al. Angiotensin I-converting enzyme inhibitory peptides in red-mold rice made by Monascus purpureus[J]. Process Biochemistry, 2009, 44(10): 1139-1143.
[38] KANG M G, KIM J H, AHN B H, et al. Characterization of new antihypertensive angiotensin I-converting enzyme inhibitory peptides from Korean traditional rice wine[J]. Journal of Microbiology and Biotechnology, 2012, 22(3): 339-342.
[39] NEMOTO Y, MATSUO Y, SHIODA K, et al. Compositional differences, antioxidant activity, angiotensin-converting enzyme inhibition, and sensory characteristics of kamaboko fermented in wheat, rice, brown rice, and soybean koji[J]. Fisheries Science, 2020, 86(3): 581-593.
[40] HU Y, SUN-WATERHOUSE D, LIU P Z, et al. Modification of rice protein with glutaminase for improved structural and sensory properties[J]. International Journal of Food Science and Technology, 2019, 54(7): 2458-2467.
[41] DONG J J, WANG S, YIN X Y, et al. Angiotensin I converting enzyme (ACE) inhibitory activity and antihypertensive effects of rice peptides[J]. Food Science and Human Wellness, 2022, 11(6): 1539-1543.
[42] DING Y H, HAN P, MA H L, et al. Process control of rice protein enzymolysis by field monitoring[J]. Current Research in Biotechnology, 2024, 7: 100171.
[43] LONG Y H, GAO Z, QIU C H, et al. Rice dreg protein as a high-quality plant-based protein resource: Characterization of structural, functional, and in vitro digestive properties[J]. Cereal Chemistry, 2024, 101(4): 785-797.
[44] SUWANNACHOT P, THAWORNCHINSOMBUT S, JONGJAREONRAK A, et al. Supplementation with rice bran hydrolysates reduces oxidative stress and improves lipid profiles in adult dogs[J]. Journal of Veterinary Medical Science, 2023, 85(7): 727-734.
[45] JUNSARA K, YUPANQUI C T, KAWEE-AI A, et al. Fortification of crude protein extract from Sung Yod and Hom rajinee rice brans in the development of functional jelly products[J]. Foods, 2023, 12(6): 1138.
[46] KUKONGVIRIVAPAN U, JAN-ON G, TUBSAKUL A, et al. Sang-Yod rice bran hydrolysates alleviate hypertension, endothelial dysfunction, vascular remodeling, and oxidative stress in nitric oxide deficient hypertensive rats[J]. Asian Pacific Journal of Tropical Biomedicine, 2021, 11(1): 10-19.
[47] ZHANG L Y, MIAO J Y, GUO J B, et al. Two novel angiotensin I-converting enzyme (ACE) inhibitory peptides from rice (Oryza sativa L.) bran protein[J]. Journal of Agricultural and Food Chemistry, 2023, 71(9): 4153-4162.
[48] WU Y, ZHANG X X, CUI H W, et al. Isolation, identification and activity analysis of antioxidant peptides from rice wine lees[J]. Journal of Food Measurement and Characterization, 2023, 18(2): 1528-1542.
[49] SU H X, FAN W L, XU Y, et al. Preparation, identification, and molecular docking of novel angiotensin-converting enzyme inhibitory peptides derived from rice-based distillers’ spent cakes[J]. Journal of the Science of Food and Agriculture, 2024, 104(11): 6506-6517.
[50] WONGSA P, YUENYONGRATTANAKORN K, PONGVACHIRINT W, et al. Improving anti-hypertensive properties of plant-based alternatives to yogurt fortified with rice protein hydrolysate[J]. Heliyon, 2022, 8(10): e11087.
[51] NGAMSUK S, HSU J L, HUANG T C. Ultrasonication of milky stage rice milk with bioactive peptides from rice bran: Its bioactivities and absorption[J]. Food and Bioprocess Technology, 2020, 13(3): 462-474.
[52] DOU B X, LIU Y, LIU Y M. Isolation and characterization of angiotensin I converting enzyme (ACE) inhibitory peptides from rice bran proteins and evaluation of activity and stability[J]. Pakistan Journal of Zoology, 2020, 52(4): 1383-1391.
[53] SHOBAKO N, OGAWA Y, ISHIKADO A, et al. A novel antihypertensive peptide identified in thermolysin-digested rice bran[J]. Molecular Nutrition & Food Research, 2018, 62(4): 1700732.
[54] SHOBAKO N, ISHIKADO A, OGAWA Y, et al. Vasorelaxant and antihypertensive effects that are dependent on the endothelial NO system exhibited by rice bran-derived tripeptide[J]. Journal of Agricultural and Food Chemistry, 2019, 67(5): 1437-1442.
[55] SUWANNAPAN O, WACHIRATTANAPONGMETEE K, THAWORNCHINSOMBUT S, et al. Angiotensin-I-converting enzyme (ACE)-inhibitory peptides from Thai jasmine rice bran protein hydrolysates[J]. International Journal of Food Science & Technology, 2020, 55(6): 2441-2450.
[56] SARRINGKARIN W, LAOKULDILOK T. Optimization of the production conditions of glutinous rice bran protein hydrolysate with antioxidative properties[J]. Chiang Mai University Journal of Natural Sciences, 2017, 16(1): 0001.
[57] PIOTROWICZ I B B, GARCÉS-RIMÓN M, MORENO-FERNÁNDEZ S, et al. Antioxidant, angiotensin-converting enzyme inhibitory properties and blood-pressure-lowering effect of rice bran protein hydrolysates[J]. Foods, 2020, 9(6): 812.
[58] SELAMASSAKUL O, LAOHAKUNJIT N, KERDCHOEUEN O, et al. Bioactive peptides from brown rice protein hydrolyzed by bromelain: Relationship between biofunctional activities and flavor characteristics[J]. Journal of Food Science, 2020, 85(3): 707-717.
[59] CÁCERES P J, PEÑAS E, MARTÍNEZ-VILLALUENGA C, et al. Development of a multifunctional yogurt-like product from germinated brown rice[J]. LWT-Food Science and Technology, 2019, 99: 306-312.
[60] AHMADIFARD N, MURUETA J H C, ABEDIAN-KENARI A, et al. Comparison the effect of three commercial enzymes for enzymatic hydrolysis of two substrates (rice bran protein concentrate and soybean protein) with SDS-PAGE[J]. Journal of Food Science and Technology, 2016, 53(2): 1279-1284.
[61] CHEN L, WANG L L, LI J K, et al. Antihypertensive potential of fermented milk: The contribution of lactic acid bacteria proteolysis system and the resultant angiotensin-converting enzyme inhibitory peptide[J]. Food & Function, 2021, 12(22): 11121-11131.
[62] JUILLARD V, LOPEZ-KLEINE L, MONNET V. Lactic acid bacteria: Proteolytic systems[M]//MCSWEENEY P L H, MCNAMARA J P. Encyclopedia of Dairy Sciences (3rd ed.). Oxford: Academic Press, 2022: 249-255.
[63] MUSTAFA M H, SOLEIMANIAN-ZAD S, SHEIKHZEINODDIN M. Characterization of a trypsin-like protease 1 produced by a probiotic Lactobacillus plantarum subsp. plantarum PTCC 1896 from skimmed milk based medium[J]. LWT-Food Science and Technology, 2020, 119: 108818.
[64] SOLIERI L, RUTELLA G S, TAGLIAZUCCHI D. Impact of non-starter lactobacilli on release of peptides with angiotensin-converting enzyme inhibitory and antioxidant activities during bovine milk fermentation[J]. Food Microbiology, 2015, 51: 108-116.
[65] YIN L B, LIU Y L, HE P, et al. Optimization of antihypertensive peptide preparation by Lactobacillus plantarum fermentation of soy whey[J]. Journal of Biobased Materials and Bioenergy, 2021, 15(2): 238-243.
[66] SINGH B P, VIJ S. Growth and bioactive peptides production potential of Lactobacillus plantarum Strain C2 in soy milk: A LC-MS/MS based revelation for peptides biofunctionality[J]. LWT-Food Science and Technology, 2017, 86: 293-301.
[67] ZAREIAN M, OSKOUEIAN E, FORGHANI B, et al. Production of a wheat-based fermented rice enriched with γ-aminobutyric acid using Lactobacillus plantarum MNZ and its antihypertensive effects in spontaneously hypertensive rats[J]. Journal of Functional Foods, 2015, 16: 194-203.
[68] PUSPITOJATI E, CAHYANTO M N, MARSONO Y, et al. Changes in amino acid composition during fermentation and its effects on the inhibitory activity of angiotensin-I converting enzyme of jack bean tempe following in vitro gastrointestinal digestion[J]. Journal of Food and Nutrition Research, 2019, 58(4): 319-327.
[69] GE H J, ZHANG Z K, XIAO J X, et al. Release of Leu-Pro-Pro from corn gluten meal by fermentation with a Lactobacillus helveticus strain[J]. Journal of the Science of Food and Agriculture, 2022, 102(3): 1095-1104.
[70] SEKINE T, NAGAI H, HAMADA-SATO N. Antihypertensive and probiotic effects of hidakakombu (Saccharina angustata) fermented by Lacticaseibacillus casei 001[J]. Foods, 2021, 10(9): 2048.
[71] PARMAR H, HATI S, SAKURE A. In vitro and in silico analysis of novel ACE-inhibitory bioactive peptides derived from fermented goat milk[J]. International Journal of Peptide Research and Therapeutics, 2018, 24(3): 441-453.
[72] WU Q, LI Y, PENG K, et al. Isolation and characterization of three antihypertension peptides from the mycelia of Ganoderma lucidum (Agaricomycetes)[J]. Journal of Agricultural and Food Chemistry, 2019, 67(29): 8149-8159.
[73] LU F, ALENYOREGE E A, OUYANG N N, et al. Simulated natural and high temperature solid-state fermentation of soybean meal: A comparative study regarding microorganisms, functional properties and structural characteristics[J]. LWT-Food Science and Technology, 2022, 159: 113125.
[74] MOAYEDI A, MORA L, ARISTOY M C, et al. ACE-inhibitory and antioxidant activities of peptide fragments obtained from tomato processing by-products fermented using Bacillus subtilis: Effect of amino acid composition and peptides molecular mass distribution[J]. Applied Biochemistry and Biotechnology, 2016, 181(1): 48-64.
[75] TAMAM B, SYAH D, SUHARTONO M T, et al. Proteomic study of bioactive peptides from tempe[J]. Journal of Bioscience and Bioengineering, 2019, 128(2): 241-248.
[76] ARDIANSYAH A. A short review: Bioactivity of fermented rice bran[J]. Journal of Oleo Science, 2021, 70(11): 1565-1574.
[77] HU J F, WANG H Y, WENG N H, et al. Novel angiotensin-converting enzyme and pancreatic lipase oligopeptide inhibitors from fermented rice bran[J]. Frontiers in Nutrition, 2022, 9: 1010005.
[78] ALAUDDIN M, SHIRAKAWA H, KOSEKI T, et al. Fermented rice bran supplementation mitigates metabolic syndrome in stroke-prone spontaneously hypertensive rats[J]. BMC Complementary and Alternative Medicine, 2016, 16(1): 442.
[79] GUO J B, LU A C, SUN Y N, et al. Purification and identification of antioxidant and angiotensin converting enzyme-inhibitory peptides from Guangdong glutinous rice wine[J]. LWT-Food Science and Technology, 2022, 169: 113953.
[80] MISHRA B K, DAS S, PRAJAPATI J B, et al. Bio-functional properties and storage study of ‘Chubitchi’- a fermented rice beverage of Garo Hills, Meghalaya[J]. Indian Journal of Traditional Knowledge, 2021, 20(2): 498-511.
[81] DALIRI E B, OFOSU F K, CHELLIAH R, et al. Untargeted metabolomics of fermented rice using UHPLC QTOF MS/MS reveals an abundance of potential antihypertensive compounds[J]. Foods, 2020, 9(8): 1007.
[82] POOJA K, RANI S, PRAKASH B. In silico approaches towards the exploration of rice bran proteins-derived angiotensin-I-converting enzyme inhibitory peptides[J]. International Journal of Food Properties, 2017, 20(2): 2178-2191.
[83] Xia X Y, Wen C X, Cao W H, et al. Screening of ACE inhibitory peptides from enzymatic hydrolysates of Pinctada martensii meat based on surface plasmon resonance technology[J]. Food Science, 2025, 46(7): 143-150.
[84] WU N, LI P, SHUANG Q, et al. Screening and molecular dynamics simulation of ACE inhibitory tripeptides derived from milk fermented with Lactobacillus delbrueckii QS306[J]. Food & Function, 2024, 15(5): 2655-2667.
Research Progress on Angiotensin-Converting Enzyme Inhibitory Peptides Derived from Rice
Hypertension is a prevalent and common chronic disease that severely endangers human life and health. Globally, approximately 3.5 billion adults present suboptimal blood pressure levels, with a notable trend of younger onset [1]. The blood pressure regulatory system comprises the autonomic nervous system, vascular tone regulation system, renin-angiotensin-aldosterone system (RAAS), renal system and other functional modules. Among these, the RAAS exerts a pivotal role in arterial blood pressure regulation and extracellular fluid homeostasis maintenance [2].
Angiotensin-converting enzyme (ACE) serves as a core component of the RAAS. Inhibition of ACE activity reduces the biosynthesis of angiotensin II (Ang II), thereby inducing vasodilation and hypotensive effects. Currently, angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers constitute the predominant first-line antihypertensive pharmacotherapies [3-4]. Although these clinical agents exhibit satisfactory tolerability, nearly 20% of patients discontinue ACEI treatment due to adverse drug reactions such as chronic cough [5]. Accordingly, it is imperative to explore safer and more efficient hypotensive strategies and develop novel ACEIs characterized by superior efficacy, minimal side effects and high biosafety.
Accumulated studies have demonstrated that bioactive peptides liberated from food proteins possess multiple physiological functions, including antioxidant, antihypertensive, antibacterial, antithrombotic and immunomodulatory activities [6-7]. Angiotensin-converting enzyme inhibitory peptides (ACEIPs) are distinguished by high biological activity, favorable biosafety and efficient in vivo metabolic characteristics, rendering them increasingly prominent in the research and development of functional foods [8]. ACEIPs and their extracts can be efficiently prepared from dietary raw materials such as milk, meat and rice via enzymatic hydrolysis and microbial fermentation. These bioactive compounds exhibit potent ACE inhibitory capacity and prominent hypotensive properties, demonstrating broad application prospects in the food industry [9-11]. Nevertheless, the production of animal-derived proteins entails excessive energy consumption and substantial greenhouse gas emissions, which conflict with contemporary environmental sustainability concepts [12]. Plant-based raw materials, particularly cereal crops represented by rice, have garnered escalating research attention for ACEIP preparation by virtue of abundant raw material reserves and eco-friendly production advantages [7,13-14]. This review systematically summarizes the regulatory mechanisms of the blood pressure homeostasis system, the hypotensive pathways of ACEIPs, as well as their structural and functional characteristics. Particular emphasis is placed on elaborating the research advances in the preparation, separation and purification of rice-derived ACEIPs through enzymatic hydrolysis and fermentation technologies. This work aims to provide theoretical foundations for the industrial development of food-originated ACEIP products and the high-value extension of the rice industrial chain.
一.Blood Pressure Regulation and Hypotensive Mechanisms
1.Regulatory Mechanisms of the Blood Pressure System
The inhibitory efficacy against ACE is primarily associated with two pivotal blood pressure regulatory axes: the renin-angiotensin system (RAS) and nitric oxide system (NOS). As illustrated in Figure 1, renin, a proteolytic enzyme synthesized by juxtaglomerular cells, catalyzes the conversion of angiotensinogen into angiotensin I (Ang I). ACE further mediates the transformation of Ang I into Ang II, which subsequently activates angiotensin II type 1 receptors (AT1R) and triggers a cascade of pathological responses including vasoconstriction, tissue fibrosis, inflammatory infiltration and reactive oxygen species (ROS) overproduction. Meanwhile, moderate levels of Ang II bind to angiotensin II type 2 receptors (AT2R) to counterbalance AT1R-mediated biological effects, thereby sustaining the physiological homeostasis of the RAS [15].
Zinc metalloprotease ACE2 is capable of degrading Ang II to generate Ang-(1-7), or directly converting Ang I into Ang-(1-9). The intermediate product Ang-(1-9) can be further hydrolyzed by ACE to produce Ang-(1-7). Functionally opposite to Ang II, Ang-(1-7) specifically couples with corresponding G-protein-coupled receptors to exert vasodilatory, anti-inflammatory and anti-fibrotic bioactivities, and is ultimately degraded by ACE into inactive Ang-(1-5) fragments [16]. Hence, ACE occupies a more dominant regulatory position than ACE2 within the RAS cascade. Both aminopeptidase A (AP-A) and aminopeptidase N (AP-N) catalyze the biotransformation of Ang II into Ang IV. Additionally, Ang IV binds to insulin-regulated aminopeptidase receptors (IRAP), resulting in adverse physiological outcomes such as vasoconstriction, inflammatory responses and ROS accumulation [17].
2.Structural Characteristics of ACEIPs
The physiological functions of ACEIPs are intrinsically correlated with their polypeptide structures, with key determinants including molecular weight, amino acid composition and sequence arrangement. The bioactivity of ACE inhibitory peptides is closely related to molecular mass. Multiple studies have verified that short-chain peptides containing 2 to 12 amino acid residues (with molecular weights generally below 3000 Da) can optimally bind to the active sites of ACE [18-19]. In most ACEIP sequences, the last three amino acid residues at the C-terminus are hydrophobic amino acids, which bind to the terminal catalytic sites of ACE to achieve targeted enzyme inhibition.
ACEIPs with proline (Pro) at the C-terminus have been validated to possess enhanced inhibitory potency and superior resistance to digestive enzyme degradation [20-21]. Hydroxyproline (Hyp) plays a critical role in the molecular interaction with the ACE active pocket, and its presence at the penultimate position of the C-terminus significantly enhances ACE inhibitory activity [22-23]. For tetrapeptide ACEIPs, specific residue combinations confer potent inhibitory effects: tyrosine (Tyr) or cysteine (Cys) at the first position, histidine (His), tryptophan (Trp) or methionine (Met) at the second position, isoleucine (Ile), leucine (Leu), valine (Val) or methionine (Met) at the third position, and tryptophan (Trp) at the fourth position [24-25]. Molecular docking analysis further clarifies the binding modes of ACEIPs. As depicted in Figure 2, the tripeptide Tyr-Ser-Lys (YSK) exerts ACE inhibitory effects mainly via hydrogen bond formation within the ACE active pocket [26].
Current research also indicates that hydrophobic amino acids at the N-terminus endow polypeptides with strong ACE inhibitory capacity, while basic amino acid residues at the N-terminus can strengthen molecular affinity to ACE and further elevate antihypertensive bioactivity [18-19,27]. Beyond the 20 canonical amino acids, non-canonical amino acids also modulate ACEIP bioactivity. For instance, citrulline (Cit) with neutral net charge increases protein hydrophobicity upon accumulation; ornithine (Orn), a typical basic amino acid, can enhance ACEIP activity when located at the N-terminus. Both non-canonical amino acids exhibit antioxidant and vascular-protective properties, which may assist in alleviating adverse physiological side effects [28-30].
3.Preparation Methods of Rice-Derived ACEIPs
Major fabrication technologies for bioactive peptides include solvent extraction, enzymatic hydrolysis, microbial fermentation, chemical synthesis and genetic engineering recombination [31]. Solvent extraction is characterized by simple principles and convenient operational procedures; in vitro synthetic approaches such as chemical synthesis and recombinant genetic engineering offer distinct advantages of short production cycles and high yield, effectively compensating for the limitations of natural peptide acquisition [32]. In contrast, enzymatic hydrolysis and microbial fermentation have evolved as mature industrial preparation methods due to high biosafety, robust biological activity and specific catalytic specificity. With the rapid advancement of biotechnology, innovative strategies for natural peptide production continue to emerge. At present, enzymatic hydrolysis and microbial fermentation represent the mainstream technical routes for ACEIP production using food-derived raw materials.
二. Preparation of Rice-Derived ACEIPs via Enzymatic Hydrolysis
1. Utilization of Different Rice Fractions
Rice contains a relatively low protein content (approximately 8%), whereas the nutritional value and health-promoting properties of rice proteins have been widely recognized [33]. Native rice proteins exhibit poor solubility and digestibility under neutral conditions, and enzymatic modification can effectively improve their functional and biological properties. Proteins isolated from rice can be hydrolyzed into abundant bioactive peptides, which exert dose-dependent ACE inhibitory effects at appropriate concentrations [34-35]. As summarized in Table 1, rice protein hydrolysates prepared via hydrolysis with multiple commercial enzyme preparations display efficient in vivo and in vitro hypotensive activities. Several typical rice-derived ACEIPs have been successfully identified, including Ile-His-Arg-Phe (IHRF), Val-Asn-Pro (VNP) and Val-Trp-Pro (VWP) [36-37].
Preliminary separation of rice protein hydrolysates can be achieved through ultrafiltration and DA201 macroporous adsorption resin, followed by two-step reversed-phase high-performance liquid chromatography (RP-HPLC) purification to obtain two high-purity tripeptides with prominent ACE inhibitory activity [37]. Six novel ACE inhibitory peptides have been separately isolated from red yeast rice and traditional Korean rice wine in previous studies [38-39]. Moreover, oral administration of fermented brown rice aqueous extracts and rice residue protein hydrolysates containing peptide components yields marked hypotensive outcomes in spontaneously hypertensive rats (SHRs) [40].
Proteins extracted from rice processing by-products present favorable biological characteristics (Table 1). Rice bran, rice residue and distillers’ grains are cost-effective and sustainable raw material sources for ACEIP production. Unhydrolyzed rice bran proteins demonstrate weaker ACE inhibitory activity compared with their enzymatic hydrolysates. Commercial protease-treated rice bran protein hydrolysates feature low relative molecular weight and potent in vitro ACE inhibitory activity. Enzymatically modified rice bran proteins can be developed as high-value functional food ingredients with superior bioavailability and absorption efficiency [51].
Novel bioactive peptides are continuously discovered from rice by-products. For example, a novel ACE inhibitory peptide with the sequences of Ile-Thr-Leu (ITL) or Leu-Thr-Ile (LTI) was purified from alkaline protease-hydrolyzed rice bran protein via size-exclusion chromatography and RP-HPLC, whose in vitro ACE inhibitory potency was comparable to that of enalapril maleate [47,52]. Multiple dipeptides and tripeptides with validated in vivo and in vitro ACE inhibitory effects have been identified in rice bran enzymatic hydrolysates [53-54]. Rice bran protein hydrolysates exhibit efficient ACE suppression capacity, with pharmacological effects analogous to the classic hypotensive drug captopril [55]. Additionally, protein hydrolysates derived from brown rice, refined rice protein and germinated brown rice also possess inherent ACE inhibitory potential [56-59].
2.Application of Enzymatic Preparations
As listed in Table 1, proteases including alkaline protease, trypsin, neutral protease and pepsin display unique cleavage sites and optimal reaction conditions, contributing to differential hypotensive performance in ACEIP preparation. Alkaline protease is the most widely adopted and high-efficiency protease in current research, which is closely associated with the poor solubility of rice proteins under acidic and neutral environments. A comparative study on the enzymatic hydrolysis efficiency of rice bran protein concentrate and soybean protein was conducted using alkaline protease, papain and commercial compound enzymes containing trypsin, thymoprotease and aminopeptidase. The results confirmed that alkaline protease possesses superior hydrolytic capacity and ACE inhibitory modification efficiency [60].
Enzymatically treated rice bran serves as an excellent natural source of bioactive peptides, showing promising therapeutic potential for hypertension and related metabolic disorders. Beyond ACE inhibitory activity, rice protein hydrolysates obtained via enzymatic degradation exhibit strong free radical scavenging capacity and reducing power. These multifunctional hydrolysates can be applied as functional food additives in nutritional foods and beverages, delivering comprehensive health benefits and satisfactory sensory quality [56-59].
三. Separation and Identification Technologies for ACEIPs
The conventional technical workflow for ACEIP separation and identification consists of three core steps: separation of rice-derived crude polypeptides using membrane separation technology, gel filtration chromatography and RP-HPLC; amino acid sequence identification via mass spectrometry; and determination of interaction sites and binding patterns through molecular docking simulation, which collectively assist the targeted separation and structural characterization of ACEIPs [26,37,47].
Innovative monitoring technologies have also been integrated into the enzymatic hydrolysis process. For instance, in-situ near-infrared spectroscopy combined with chemometric algorithms enables real-time detection of ACE inhibitory activity during dual-enzyme hydrolysis of rice-derived substrates. This integrated monitoring system can precisely regulate the hydrolysis termination point and adaptive transition parameters, thereby improving overall enzymatic hydrolysis efficiency [42]. Surface plasmon resonance and other biophysical technologies can simulate enzymatic hydrolysis and fermentation conditions to screen potential bioactive peptides, facilitate structural characterization and mechanism elucidation, and provide theoretical guidance for the functional prediction of ACE inhibitory peptides.
In silico screening strategies are increasingly applied in ACEIP research. By comparing acquired peptide sequences with published polypeptide databases from plant, animal and other food sources, bioinformatics databases such as BIOPEP and computer-aided matching tools can rapidly screen candidate bioactive peptides, whose actual biological activity is further verified through in vitro experiments to explore novel ACE inhibitory components [82-83]. Furthermore, systematic assessment and predictive analysis of the physicochemical properties, primary structures, toxicological risks and allergenicity of most ACE inhibitory peptides can be performed, so as to comprehensively guarantee the safety of final functional food products [84]. The combined application of these advanced separation and identification technologies greatly promotes the basic research and industrial development of rice-derived hypotensive functional foods and other bioactive food products.
四. Research Prospects and Limitations of Rice-Derived ACEIPs
The production of ACEIPs from rice and its processing by-products via enzymatic hydrolysis, microbial fermentation or combined biotechnological strategies presents enormous development potential. Consumers have gradually recognized the unique functional advantages and nutritional value of rice-derived functional products. Nevertheless, compared with animal-derived and other plant-derived bioactive peptides, research on rice-based ACEIPs prepared by enzymatic hydrolysis and fermentation remains relatively limited, leaving substantial room for in-depth exploration and industrial application.
The structure-activity relationship of ACEIPs and their functional regulatory mechanisms require further systematic investigation. In particular, in vivo metabolic pathways and clinical evidence of fermented rice-derived ACEIPs are still insufficient. Future research on the bioactivity of ACEIPs is expected to rely on multi-omics technologies including genomics and metabolomics to clarify their biosynthesis mechanisms and structural contributions to physiological functions. Transcriptomics and other molecular biological approaches, combined with computational simulation software, can be utilized to analyze the interactive relationships among fermentative microorganisms, optimize the targeted biosynthesis of rice-derived bioactive peptides, and elucidate their intracellular metabolism and transmembrane transport mechanisms.
Further in-depth research is urgently required to enhance peptide production efficiency and oral bioavailability, evaluate the in vivo bioaccessibility and long-term hypotensive efficacy of rice-derived ACEIPs. Collectively, ongoing mechanistic and applied research will provide solid theoretical and technical support for the innovative development and clinical auxiliary application of health care products and adjuvant antihypertensive agents based on rice bioactive peptides.
References:
[1] DZAU V J, HODGKINSON C P. Precision hypertension[J]. Hypertension, 2024, 81(4): 702-708.
[2] OLFAT A, MOSTAGHIM T, SHAHRIARI S, et al. Extraction of bioactive compounds of Hypnea flagelliformis by ultrasound-assisted extraction coupled with natural deep eutectic solvent and enzyme inhibitory activity[J]. Algal Research, 2024, 78: 103388.
[3] OPARIL S, ACELAJADO M C, BAKRIS G L, et al. Hypertension[J]. Nature Reviews Disease Primers, 2018, 4: 18014.
[4] WANG J W, YE Y Y, CHEN X F, et al. Diverse trends in antihypertensive medication usage among U.S. adults with hypertension by socioeconomic status and comorbidities, 1999—2020[J]. Blood Pressure, 2025, 34(1): 2506081.
[5] FABIAN I M, MADDOX K, ROBICHEAUX C, et al. Stevens-Johnson syndrome from combined allopurinol and angiotensin-converting enzyme inhibitors: A narrative review[J]. Cureus, 2024, 16(1): e51899.
[6] VILLANUEVA A, RIVERO-PINO F, MARTIN M E, et al. Identification of the bioavailable peptidome of chia protein hydrolysate and the in silico evaluation of its antioxidant and ACE inhibitory potential[J]. Journal of Agricultural and Food Chemistry, 2024, 72(6): 3189-3199.
[7] RAMAKRISHNAN V V, HOSSAIN A, DAVE D, et al. Salmon processing discards: a potential source of bioactive peptides-a review[J]. Food Production, Processing and Nutrition, 2024, 6(1): 22.
[8] XIANG L, QIU Z C, ZHAO R J, et al. Advancement and prospects of production, transport, functional activity and structure-activity relationship of food-derived angiotensin converting enzyme (ACE) inhibitory peptides[J]. Critical Reviews in Food Science and Nutrition, 2023, 63(10): 1437-1463.
[9] HERES A, YOKOYAMA I, GALLEGO M, et al. Antihypertensive potential of sweet Ala-Ala dipeptide and its quantitation in dry-cured ham at different processing conditions[J]. Journal of Functional Foods, 2021, 87: 104818.
[10] SHANMUGAM V P, KAPILA S, KEMGANG T S, et al. Isolation and characterization of angiotensin converting enzyme inhibitory peptide from buffalo casein[J]. International Journal of Peptide Research and Therapeutics, 2021, 27(2): 1481-1491.
[11] KAISER S, MARTIN M, LUNOW D, et al. Tryptophan-containing dipeptides are bioavailable and inhibit plasma human angiotensin-converting enzyme in vivo[J]. International Dairy Journal, 2016, 52: 107-114.
[12] ZHANG D J, JIANG K, LUO H, et al. Replacing animal proteins with plant proteins: Is this a way to improve quality and functional properties of hybrid cheeses and cheese analogs?[J]. Comprehensive Reviews in Food Science and Food Safety, 2024, 23(1): e13262.
[13] PREKUMAR J, MALINI M, JOSHY A. A critical review on food protein-derived antihypertensive peptides[J]. Drug Invention Today, 2019, 12(3): 474-479.
[14] CHUKWUMA C I. Antioxidative, metabolic and vascular medicinal potentials of natural products in the non-edible wastes of fruits belonging to the Citrus and Prunus genera: A review[J]. Plants, 2024, 13(2): 191.
[15] FARIA-COSTA G, LEITE-MOREIRA A, HENRIQUES-COELHO T. Efeitos cardiovasculares do receptor tipo 2 da angiotensina[J]. Revista Portuguesa de Cardiologia, 2014, 33(7/8): 439-449.
[16] METLOCK F E, HINNEH T, BENJASIRISAN C, et al. Impact of social determinants of health on hypertension outcomes: A systematic review[J]. Hypertension, 2024, 81(8): 1675-1700.
[17] HALLBERG M, LARHED M. From angiotensin IV to small peptidemimetics inhibiting insulin-regulated aminopeptidase[J]. Frontiers in Pharmacology, 2020, 11: 590855.
[18] SUN S Q, XU X T, SUN X, et al. Preparation and identification of ACE inhibitory peptides from the marine macroalga Ulva intestinalis[J]. Marine Drugs, 2019, 17(3): 179.
[19] TOOPCHAM T, MES J J, WICHERS H J, et al. Bioavailability of angiotensin I-converting enzyme (ACE) inhibitory peptides derived from Virgibacillus halodenitrificans SK1-3-7 proteinases hydrolyzed tilapia muscle proteins[J]. Food Chemistry, 2017, 220: 190-197.
[20] MIRALLES B, AMIGO L, RECIO I. Critical review and perspectives on food-derived antihypertensive peptides[J]. Journal of Agricultural and Food Chemistry, 2018, 66(36): 9384-9390.
[21] JAO C L, HUANG S L, HSU K C. Angiotensin I-converting enzyme inhibitory peptides: Inhibition mode, bioavailability, and antihypertensive effects[J]. BioMedicine, 2012, 2(4): 130-136.
[22] CIAU-SOLÍS N, RODRÍGUEZ-CANTO W, FERNÁNDEZ-MARTÍNEZ L, et al. Inhibitory activity of angiotensin-I converting enzyme (ACE-I) from partially purified Phaseolus lunatus peptide fractions[J]. Process Biochemistry, 2024, 139: 44-50.
[23] TAGA Y, HAYASHIDA O, ASHOUR A, et al. Characterization of angiotensin-converting enzyme inhibitory activity of X-hyp-gly-type tripeptides: Importance of collagen-specific prolyl hydroxylation[J]. Journal of Agricultural and Food Chemistry, 2018, 66(33): 8737-8743.
[24] LIU C, ZHANG Z J, SHANG Y T, et al. Progress in the preparation, identification and biological activity of walnut peptides[J]. Journal of Future Foods, 2024, 4(3): 205-220.
[25] XIANG L, ZHENG Z J, GUO X J, et al. Two novel angiotensin I-converting enzyme inhibitory peptides from garlic protein: In silico screening, stability, antihypertensive effects in vivo and underlying mechanisms[J]. Food Chemistry, 2024, 435: 137537.
[26] WANG X M, CHEN H X, FU X G, et al. A novel antioxidant and ACE inhibitory peptide from rice bran protein: Biochemical characterization and molecular docking study[J]. LWT-Food Science and Technology, 2017, 75: 93-99.
[27] LEE S Y, HUR S J. Antihypertensive peptides from animal products, marine organisms, and plants[J]. Food Chemistry, 2017, 228: 506-517.
[28] Chen S R, Wei T, Li Q, et al. Process optimization and dynamic analysis of ACE inhibitory peptides produced by fermenting buffalo milk with Lactobacillus plantarum Strain B[J]. Cereals & Oils, 2024, 37(12): 89-96.
[29] LI H J, WANG Y S, WANG Y N, et al. Mechanical study of alisol B23-acetate on methionine and choline deficient diet-induced nonalcoholic steatohepatitis based on untargeted metabolomics[J]. Biomedical Chromatography, 2024, 38(1): e5763.
[30] BUTTERWORTH R F, CANBAY A. Hepatoprotection by L-ornithine L-aspartate in non-alcoholic fatty liver disease[J]. Digestive Diseases, 2019, 37(1): 63-68.
[31] MONDAL S, DAS S, NANDI A K. A review on recent advances in polymer and peptide hydrogels[J]. Soft Matter, 2020, 16(6): 1404-1454.
[32] FENG L, WANG Y, YANG J, et al. Overview of the preparation method, structure and function, and application of natural peptides and polypeptides[J]. Biomedecine & Pharmacotherapie, 2022, 153: 113493.
[33] TYAGI A, CHELLIAH R, BANAN-MWINE DALIRI E, et al. Antioxidant activities of novel peptides from Limosilactobacillus reuteri fermented brown rice: A combined in vitro and in silico study[J]. Food Chemistry, 2023, 404(Pt B): 134747.
[34] DALIRI E B, LEE B H, OH D H. Current trends and perspectives of bioactive peptides[J]. Critical Reviews in Food Science and Nutrition, 2018, 58(13): 2273-2284.
[35] KONTANI N, OMAE R, KAGEBAYASHI T, et al. Characterization of Ile-His-Arg-Phe, a novel rice-derived vasorelaxing peptide with hypotensive and anorexigenic activities[J]. Molecular Nutrition & Food Research, 2014, 58(2): 359-364.
[36] KUMAR A, SINGH N, JOSHI R. Evaluation of antioxidant and ACE inhibitory peptides in rice bran protein hydrolysate using non-targeted UHPLC-QTOF-IMS profiling, MALDI-TOF-TOF-MS/MS, and molecular docking study[J]. International Journal of Food Science & Technology, 2024, 59(6): 4300-4314.
[37] KUBA M, TANAKA K, SESOKO M, et al. Angiotensin I-converting enzyme inhibitory peptides in red-mold rice made by Monascus purpureus[J]. Process Biochemistry, 2009, 44(10): 1139-1143.
[38] KANG M G, KIM J H, AHN B H, et al. Characterization of new antihypertensive angiotensin I-converting enzyme inhibitory peptides from Korean traditional rice wine[J]. Journal of Microbiology and Biotechnology, 2012, 22(3): 339-342.
[39] NEMOTO Y, MATSUO Y, SHIODA K, et al. Compositional differences, antioxidant activity, angiotensin-converting enzyme inhibition, and sensory characteristics of kamaboko fermented in wheat, rice, brown rice, and soybean koji[J]. Fisheries Science, 2020, 86(3): 581-593.
[40] HU Y, SUN-WATERHOUSE D, LIU P Z, et al. Modification of rice protein with glutaminase for improved structural and sensory properties[J]. International Journal of Food Science and Technology, 2019, 54(7): 2458-2467.
[41] DONG J J, WANG S, YIN X Y, et al. Angiotensin I converting enzyme (ACE) inhibitory activity and antihypertensive effects of rice peptides[J]. Food Science and Human Wellness, 2022, 11(6): 1539-1543.
[42] DING Y H, HAN P, MA H L, et al. Process control of rice protein enzymolysis by field monitoring[J]. Current Research in Biotechnology, 2024, 7: 100171.
[43] LONG Y H, GAO Z, QIU C H, et al. Rice dreg protein as a high-quality plant-based protein resource: Characterization of structural, functional, and in vitro digestive properties[J]. Cereal Chemistry, 2024, 101(4): 785-797.
[44] SUWANNACHOT P, THAWORNCHINSOMBUT S, JONGJAREONRAK A, et al. Supplementation with rice bran hydrolysates reduces oxidative stress and improves lipid profiles in adult dogs[J]. Journal of Veterinary Medical Science, 2023, 85(7): 727-734.
[45] JUNSARA K, YUPANQUI C T, KAWEE-AI A, et al. Fortification of crude protein extract from Sung Yod and Hom rajinee rice brans in the development of functional jelly products[J]. Foods, 2023, 12(6): 1138.
[46] KUKONGVIRIVAPAN U, JAN-ON G, TUBSAKUL A, et al. Sang-Yod rice bran hydrolysates alleviate hypertension, endothelial dysfunction, vascular remodeling, and oxidative stress in nitric oxide deficient hypertensive rats[J]. Asian Pacific Journal of Tropical Biomedicine, 2021, 11(1): 10-19.
[47] ZHANG L Y, MIAO J Y, GUO J B, et al. Two novel angiotensin I-converting enzyme (ACE) inhibitory peptides from rice (Oryza sativa L.) bran protein[J]. Journal of Agricultural and Food Chemistry, 2023, 71(9): 4153-4162.
[48] WU Y, ZHANG X X, CUI H W, et al. Isolation, identification and activity analysis of antioxidant peptides from rice wine lees[J]. Journal of Food Measurement and Characterization, 2023, 18(2): 1528-1542.
[49] SU H X, FAN W L, XU Y, et al. Preparation, identification, and molecular docking of novel angiotensin-converting enzyme inhibitory peptides derived from rice-based distillers’ spent cakes[J]. Journal of the Science of Food and Agriculture, 2024, 104(11): 6506-6517.
[50] WONGSA P, YUENYONGRATTANAKORN K, PONGVACHIRINT W, et al. Improving anti-hypertensive properties of plant-based alternatives to yogurt fortified with rice protein hydrolysate[J]. Heliyon, 2022, 8(10): e11087.
[51] NGAMSUK S, HSU J L, HUANG T C. Ultrasonication of milky stage rice milk with bioactive peptides from rice bran: Its bioactivities and absorption[J]. Food and Bioprocess Technology, 2020, 13(3): 462-474.
[52] DOU B X, LIU Y, LIU Y M. Isolation and characterization of angiotensin I converting enzyme (ACE) inhibitory peptides from rice bran proteins and evaluation of activity and stability[J]. Pakistan Journal of Zoology, 2020, 52(4): 1383-1391.
[53] SHOBAKO N, OGAWA Y, ISHIKADO A, et al. A novel antihypertensive peptide identified in thermolysin-digested rice bran[J]. Molecular Nutrition & Food Research, 2018, 62(4): 1700732.
[54] SHOBAKO N, ISHIKADO A, OGAWA Y, et al. Vasorelaxant and antihypertensive effects that are dependent on the endothelial NO system exhibited by rice bran-derived tripeptide[J]. Journal of Agricultural and Food Chemistry, 2019, 67(5): 1437-1442.
[55] SUWANNAPAN O, WACHIRATTANAPONGMETEE K, THAWORNCHINSOMBUT S, et al. Angiotensin-I-converting enzyme (ACE)-inhibitory peptides from Thai jasmine rice bran protein hydrolysates[J]. International Journal of Food Science & Technology, 2020, 55(6): 2441-2450.
[56] SARRINGKARIN W, LAOKULDILOK T. Optimization of the production conditions of glutinous rice bran protein hydrolysate with antioxidative properties[J]. Chiang Mai University Journal of Natural Sciences, 2017, 16(1): 0001.
[57] PIOTROWICZ I B B, GARCÉS-RIMÓN M, MORENO-FERNÁNDEZ S, et al. Antioxidant, angiotensin-converting enzyme inhibitory properties and blood-pressure-lowering effect of rice bran protein hydrolysates[J]. Foods, 2020, 9(6): 812.
[58] SELAMASSAKUL O, LAOHAKUNJIT N, KERDCHOEUEN O, et al. Bioactive peptides from brown rice protein hydrolyzed by bromelain: Relationship between biofunctional activities and flavor characteristics[J]. Journal of Food Science, 2020, 85(3): 707-717.
[59] CÁCERES P J, PEÑAS E, MARTÍNEZ-VILLALUENGA C, et al. Development of a multifunctional yogurt-like product from germinated brown rice[J]. LWT-Food Science and Technology, 2019, 99: 306-312.
[60] AHMADIFARD N, MURUETA J H C, ABEDIAN-KENARI A, et al. Comparison the effect of three commercial enzymes for enzymatic hydrolysis of two substrates (rice bran protein concentrate and soybean protein) with SDS-PAGE[J]. Journal of Food Science and Technology, 2016, 53(2): 1279-1284.
[61] CHEN L, WANG L L, LI J K, et al. Antihypertensive potential of fermented milk: The contribution of lactic acid bacteria proteolysis system and the resultant angiotensin-converting enzyme inhibitory peptide[J]. Food & Function, 2021, 12(22): 11121-11131.
[62] JUILLARD V, LOPEZ-KLEINE L, MONNET V. Lactic acid bacteria: Proteolytic systems[M]//MCSWEENEY P L H, MCNAMARA J P. Encyclopedia of Dairy Sciences (3rd ed.). Oxford: Academic Press, 2022: 249-255.
[63] MUSTAFA M H, SOLEIMANIAN-ZAD S, SHEIKHZEINODDIN M. Characterization of a trypsin-like protease 1 produced by a probiotic Lactobacillus plantarum subsp. plantarum PTCC 1896 from skimmed milk based medium[J]. LWT-Food Science and Technology, 2020, 119: 108818.
[64] SOLIERI L, RUTELLA G S, TAGLIAZUCCHI D. Impact of non-starter lactobacilli on release of peptides with angiotensin-converting enzyme inhibitory and antioxidant activities during bovine milk fermentation[J]. Food Microbiology, 2015, 51: 108-116.
[65] YIN L B, LIU Y L, HE P, et al. Optimization of antihypertensive peptide preparation by Lactobacillus plantarum fermentation of soy whey[J]. Journal of Biobased Materials and Bioenergy, 2021, 15(2): 238-243.
[66] SINGH B P, VIJ S. Growth and bioactive peptides production potential of Lactobacillus plantarum Strain C2 in soy milk: A LC-MS/MS based revelation for peptides biofunctionality[J]. LWT-Food Science and Technology, 2017, 86: 293-301.
[67] ZAREIAN M, OSKOUEIAN E, FORGHANI B, et al. Production of a wheat-based fermented rice enriched with γ-aminobutyric acid using Lactobacillus plantarum MNZ and its antihypertensive effects in spontaneously hypertensive rats[J]. Journal of Functional Foods, 2015, 16: 194-203.
[68] PUSPITOJATI E, CAHYANTO M N, MARSONO Y, et al. Changes in amino acid composition during fermentation and its effects on the inhibitory activity of angiotensin-I converting enzyme of jack bean tempe following in vitro gastrointestinal digestion[J]. Journal of Food and Nutrition Research, 2019, 58(4): 319-327.
[69] GE H J, ZHANG Z K, XIAO J X, et al. Release of Leu-Pro-Pro from corn gluten meal by fermentation with a Lactobacillus helveticus strain[J]. Journal of the Science of Food and Agriculture, 2022, 102(3): 1095-1104.
[70] SEKINE T, NAGAI H, HAMADA-SATO N. Antihypertensive and probiotic effects of hidakakombu (Saccharina angustata) fermented by Lacticaseibacillus casei 001[J]. Foods, 2021, 10(9): 2048.
[71] PARMAR H, HATI S, SAKURE A. In vitro and in silico analysis of novel ACE-inhibitory bioactive peptides derived from fermented goat milk[J]. International Journal of Peptide Research and Therapeutics, 2018, 24(3): 441-453.
[72] WU Q, LI Y, PENG K, et al. Isolation and characterization of three antihypertension peptides from the mycelia of Ganoderma lucidum (Agaricomycetes)[J]. Journal of Agricultural and Food Chemistry, 2019, 67(29): 8149-8159.
[73] LU F, ALENYOREGE E A, OUYANG N N, et al. Simulated natural and high temperature solid-state fermentation of soybean meal: A comparative study regarding microorganisms, functional properties and structural characteristics[J]. LWT-Food Science and Technology, 2022, 159: 113125.
[74] MOAYEDI A, MORA L, ARISTOY M C, et al. ACE-inhibitory and antioxidant activities of peptide fragments obtained from tomato processing by-products fermented using Bacillus subtilis: Effect of amino acid composition and peptides molecular mass distribution[J]. Applied Biochemistry and Biotechnology, 2016, 181(1): 48-64.
[75] TAMAM B, SYAH D, SUHARTONO M T, et al. Proteomic study of bioactive peptides from tempe[J]. Journal of Bioscience and Bioengineering, 2019, 128(2): 241-248.
[76] ARDIANSYAH A. A short review: Bioactivity of fermented rice bran[J]. Journal of Oleo Science, 2021, 70(11): 1565-1574.
[77] HU J F, WANG H Y, WENG N H, et al. Novel angiotensin-converting enzyme and pancreatic lipase oligopeptide inhibitors from fermented rice bran[J]. Frontiers in Nutrition, 2022, 9: 1010005.
[78] ALAUDDIN M, SHIRAKAWA H, KOSEKI T, et al. Fermented rice bran supplementation mitigates metabolic syndrome in stroke-prone spontaneously hypertensive rats[J]. BMC Complementary and Alternative Medicine, 2016, 16(1): 442.
[79] GUO J B, LU A C, SUN Y N, et al. Purification and identification of antioxidant and angiotensin converting enzyme-inhibitory peptides from Guangdong glutinous rice wine[J]. LWT-Food Science and Technology, 2022, 169: 113953.
[80] MISHRA B K, DAS S, PRAJAPATI J B, et al. Bio-functional properties and storage study of ‘Chubitchi’- a fermented rice beverage of Garo Hills, Meghalaya[J]. Indian Journal of Traditional Knowledge, 2021, 20(2): 498-511.
[81] DALIRI E B, OFOSU F K, CHELLIAH R, et al. Untargeted metabolomics of fermented rice using UHPLC QTOF MS/MS reveals an abundance of potential antihypertensive compounds[J]. Foods, 2020, 9(8): 1007.
[82] POOJA K, RANI S, PRAKASH B. In silico approaches towards the exploration of rice bran proteins-derived angiotensin-I-converting enzyme inhibitory peptides[J]. International Journal of Food Properties, 2017, 20(2): 2178-2191.
[83] Xia X Y, Wen C X, Cao W H, et al. Screening of ACE inhibitory peptides from enzymatic hydrolysates of Pinctada martensii meat based on surface plasmon resonance technology[J]. Food Science, 2025, 46(7): 143-150.
[84] WU N, LI P, SHUANG Q, et al. Screening and molecular dynamics simulation of ACE inhibitory tripeptides derived from milk fermented with Lactobacillus delbrueckii QS306[J]. Food & Function, 2024, 15(5): 2655-2667.
Research Progress on Angiotensin-Converting Enzyme Inhibitory Peptides Derived from Rice
Hypertension is a prevalent and common chronic disease that severely endangers human life and health. Globally, approximately 3.5 billion adults present suboptimal blood pressure levels, with a notable trend of younger onset [1]. The blood pressure regulatory system comprises the autonomic nervous system, vascular tone regulation system, renin-angiotensin-aldosterone system (RAAS), renal system and other functional modules. Among these, the RAAS exerts a pivotal role in arterial blood pressure regulation and extracellular fluid homeostasis maintenance [2].
Angiotensin-converting enzyme (ACE) serves as a core component of the RAAS. Inhibition of ACE activity reduces the biosynthesis of angiotensin II (Ang II), thereby inducing vasodilation and hypotensive effects. Currently, angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers constitute the predominant first-line antihypertensive pharmacotherapies [3-4]. Although these clinical agents exhibit satisfactory tolerability, nearly 20% of patients discontinue ACEI treatment due to adverse drug reactions such as chronic cough [5]. Accordingly, it is imperative to explore safer and more efficient hypotensive strategies and develop novel ACEIs characterized by superior efficacy, minimal side effects and high biosafety.
Accumulated studies have demonstrated that bioactive peptides liberated from food proteins possess multiple physiological functions, including antioxidant, antihypertensive, antibacterial, antithrombotic and immunomodulatory activities [6-7]. Angiotensin-converting enzyme inhibitory peptides (ACEIPs) are distinguished by high biological activity, favorable biosafety and efficient in vivo metabolic characteristics, rendering them increasingly prominent in the research and development of functional foods [8]. ACEIPs and their extracts can be efficiently prepared from dietary raw materials such as milk, meat and rice via enzymatic hydrolysis and microbial fermentation. These bioactive compounds exhibit potent ACE inhibitory capacity and prominent hypotensive properties, demonstrating broad application prospects in the food industry [9-11]. Nevertheless, the production of animal-derived proteins entails excessive energy consumption and substantial greenhouse gas emissions, which conflict with contemporary environmental sustainability concepts [12]. Plant-based raw materials, particularly cereal crops represented by rice, have garnered escalating research attention for ACEIP preparation by virtue of abundant raw material reserves and eco-friendly production advantages [7,13-14]. This review systematically summarizes the regulatory mechanisms of the blood pressure homeostasis system, the hypotensive pathways of ACEIPs, as well as their structural and functional characteristics. Particular emphasis is placed on elaborating the research advances in the preparation, separation and purification of rice-derived ACEIPs through enzymatic hydrolysis and fermentation technologies. This work aims to provide theoretical foundations for the industrial development of food-originated ACEIP products and the high-value extension of the rice industrial chain.
一.Blood Pressure Regulation and Hypotensive Mechanisms
1.Regulatory Mechanisms of the Blood Pressure System
The inhibitory efficacy against ACE is primarily associated with two pivotal blood pressure regulatory axes: the renin-angiotensin system (RAS) and nitric oxide system (NOS). As illustrated in Figure 1, renin, a proteolytic enzyme synthesized by juxtaglomerular cells, catalyzes the conversion of angiotensinogen into angiotensin I (Ang I). ACE further mediates the transformation of Ang I into Ang II, which subsequently activates angiotensin II type 1 receptors (AT1R) and triggers a cascade of pathological responses including vasoconstriction, tissue fibrosis, inflammatory infiltration and reactive oxygen species (ROS) overproduction. Meanwhile, moderate levels of Ang II bind to angiotensin II type 2 receptors (AT2R) to counterbalance AT1R-mediated biological effects, thereby sustaining the physiological homeostasis of the RAS [15].
Zinc metalloprotease ACE2 is capable of degrading Ang II to generate Ang-(1-7), or directly converting Ang I into Ang-(1-9). The intermediate product Ang-(1-9) can be further hydrolyzed by ACE to produce Ang-(1-7). Functionally opposite to Ang II, Ang-(1-7) specifically couples with corresponding G-protein-coupled receptors to exert vasodilatory, anti-inflammatory and anti-fibrotic bioactivities, and is ultimately degraded by ACE into inactive Ang-(1-5) fragments [16]. Hence, ACE occupies a more dominant regulatory position than ACE2 within the RAS cascade. Both aminopeptidase A (AP-A) and aminopeptidase N (AP-N) catalyze the biotransformation of Ang II into Ang IV. Additionally, Ang IV binds to insulin-regulated aminopeptidase receptors (IRAP), resulting in adverse physiological outcomes such as vasoconstriction, inflammatory responses and ROS accumulation [17].
2.Structural Characteristics of ACEIPs
The physiological functions of ACEIPs are intrinsically correlated with their polypeptide structures, with key determinants including molecular weight, amino acid composition and sequence arrangement. The bioactivity of ACE inhibitory peptides is closely related to molecular mass. Multiple studies have verified that short-chain peptides containing 2 to 12 amino acid residues (with molecular weights generally below 3000 Da) can optimally bind to the active sites of ACE [18-19]. In most ACEIP sequences, the last three amino acid residues at the C-terminus are hydrophobic amino acids, which bind to the terminal catalytic sites of ACE to achieve targeted enzyme inhibition.
ACEIPs with proline (Pro) at the C-terminus have been validated to possess enhanced inhibitory potency and superior resistance to digestive enzyme degradation [20-21]. Hydroxyproline (Hyp) plays a critical role in the molecular interaction with the ACE active pocket, and its presence at the penultimate position of the C-terminus significantly enhances ACE inhibitory activity [22-23]. For tetrapeptide ACEIPs, specific residue combinations confer potent inhibitory effects: tyrosine (Tyr) or cysteine (Cys) at the first position, histidine (His), tryptophan (Trp) or methionine (Met) at the second position, isoleucine (Ile), leucine (Leu), valine (Val) or methionine (Met) at the third position, and tryptophan (Trp) at the fourth position [24-25]. Molecular docking analysis further clarifies the binding modes of ACEIPs. As depicted in Figure 2, the tripeptide Tyr-Ser-Lys (YSK) exerts ACE inhibitory effects mainly via hydrogen bond formation within the ACE active pocket [26].
Current research also indicates that hydrophobic amino acids at the N-terminus endow polypeptides with strong ACE inhibitory capacity, while basic amino acid residues at the N-terminus can strengthen molecular affinity to ACE and further elevate antihypertensive bioactivity [18-19,27]. Beyond the 20 canonical amino acids, non-canonical amino acids also modulate ACEIP bioactivity. For instance, citrulline (Cit) with neutral net charge increases protein hydrophobicity upon accumulation; ornithine (Orn), a typical basic amino acid, can enhance ACEIP activity when located at the N-terminus. Both non-canonical amino acids exhibit antioxidant and vascular-protective properties, which may assist in alleviating adverse physiological side effects [28-30].
3.Preparation Methods of Rice-Derived ACEIPs
Major fabrication technologies for bioactive peptides include solvent extraction, enzymatic hydrolysis, microbial fermentation, chemical synthesis and genetic engineering recombination [31]. Solvent extraction is characterized by simple principles and convenient operational procedures; in vitro synthetic approaches such as chemical synthesis and recombinant genetic engineering offer distinct advantages of short production cycles and high yield, effectively compensating for the limitations of natural peptide acquisition [32]. In contrast, enzymatic hydrolysis and microbial fermentation have evolved as mature industrial preparation methods due to high biosafety, robust biological activity and specific catalytic specificity. With the rapid advancement of biotechnology, innovative strategies for natural peptide production continue to emerge. At present, enzymatic hydrolysis and microbial fermentation represent the mainstream technical routes for ACEIP production using food-derived raw materials.
二. Preparation of Rice-Derived ACEIPs via Enzymatic Hydrolysis
1. Utilization of Different Rice Fractions
Rice contains a relatively low protein content (approximately 8%), whereas the nutritional value and health-promoting properties of rice proteins have been widely recognized [33]. Native rice proteins exhibit poor solubility and digestibility under neutral conditions, and enzymatic modification can effectively improve their functional and biological properties. Proteins isolated from rice can be hydrolyzed into abundant bioactive peptides, which exert dose-dependent ACE inhibitory effects at appropriate concentrations [34-35]. As summarized in Table 1, rice protein hydrolysates prepared via hydrolysis with multiple commercial enzyme preparations display efficient in vivo and in vitro hypotensive activities. Several typical rice-derived ACEIPs have been successfully identified, including Ile-His-Arg-Phe (IHRF), Val-Asn-Pro (VNP) and Val-Trp-Pro (VWP) [36-37].
Preliminary separation of rice protein hydrolysates can be achieved through ultrafiltration and DA201 macroporous adsorption resin, followed by two-step reversed-phase high-performance liquid chromatography (RP-HPLC) purification to obtain two high-purity tripeptides with prominent ACE inhibitory activity [37]. Six novel ACE inhibitory peptides have been separately isolated from red yeast rice and traditional Korean rice wine in previous studies [38-39]. Moreover, oral administration of fermented brown rice aqueous extracts and rice residue protein hydrolysates containing peptide components yields marked hypotensive outcomes in spontaneously hypertensive rats (SHRs) [40].
Proteins extracted from rice processing by-products present favorable biological characteristics (Table 1). Rice bran, rice residue and distillers’ grains are cost-effective and sustainable raw material sources for ACEIP production. Unhydrolyzed rice bran proteins demonstrate weaker ACE inhibitory activity compared with their enzymatic hydrolysates. Commercial protease-treated rice bran protein hydrolysates feature low relative molecular weight and potent in vitro ACE inhibitory activity. Enzymatically modified rice bran proteins can be developed as high-value functional food ingredients with superior bioavailability and absorption efficiency [51].
Novel bioactive peptides are continuously discovered from rice by-products. For example, a novel ACE inhibitory peptide with the sequences of Ile-Thr-Leu (ITL) or Leu-Thr-Ile (LTI) was purified from alkaline protease-hydrolyzed rice bran protein via size-exclusion chromatography and RP-HPLC, whose in vitro ACE inhibitory potency was comparable to that of enalapril maleate [47,52]. Multiple dipeptides and tripeptides with validated in vivo and in vitro ACE inhibitory effects have been identified in rice bran enzymatic hydrolysates [53-54]. Rice bran protein hydrolysates exhibit efficient ACE suppression capacity, with pharmacological effects analogous to the classic hypotensive drug captopril [55]. Additionally, protein hydrolysates derived from brown rice, refined rice protein and germinated brown rice also possess inherent ACE inhibitory potential [56-59].
2.Application of Enzymatic Preparations
As listed in Table 1, proteases including alkaline protease, trypsin, neutral protease and pepsin display unique cleavage sites and optimal reaction conditions, contributing to differential hypotensive performance in ACEIP preparation. Alkaline protease is the most widely adopted and high-efficiency protease in current research, which is closely associated with the poor solubility of rice proteins under acidic and neutral environments. A comparative study on the enzymatic hydrolysis efficiency of rice bran protein concentrate and soybean protein was conducted using alkaline protease, papain and commercial compound enzymes containing trypsin, thymoprotease and aminopeptidase. The results confirmed that alkaline protease possesses superior hydrolytic capacity and ACE inhibitory modification efficiency [60].
Enzymatically treated rice bran serves as an excellent natural source of bioactive peptides, showing promising therapeutic potential for hypertension and related metabolic disorders. Beyond ACE inhibitory activity, rice protein hydrolysates obtained via enzymatic degradation exhibit strong free radical scavenging capacity and reducing power. These multifunctional hydrolysates can be applied as functional food additives in nutritional foods and beverages, delivering comprehensive health benefits and satisfactory sensory quality [56-59].
三. Separation and Identification Technologies for ACEIPs
The conventional technical workflow for ACEIP separation and identification consists of three core steps: separation of rice-derived crude polypeptides using membrane separation technology, gel filtration chromatography and RP-HPLC; amino acid sequence identification via mass spectrometry; and determination of interaction sites and binding patterns through molecular docking simulation, which collectively assist the targeted separation and structural characterization of ACEIPs [26,37,47].
Innovative monitoring technologies have also been integrated into the enzymatic hydrolysis process. For instance, in-situ near-infrared spectroscopy combined with chemometric algorithms enables real-time detection of ACE inhibitory activity during dual-enzyme hydrolysis of rice-derived substrates. This integrated monitoring system can precisely regulate the hydrolysis termination point and adaptive transition parameters, thereby improving overall enzymatic hydrolysis efficiency [42]. Surface plasmon resonance and other biophysical technologies can simulate enzymatic hydrolysis and fermentation conditions to screen potential bioactive peptides, facilitate structural characterization and mechanism elucidation, and provide theoretical guidance for the functional prediction of ACE inhibitory peptides.
In silico screening strategies are increasingly applied in ACEIP research. By comparing acquired peptide sequences with published polypeptide databases from plant, animal and other food sources, bioinformatics databases such as BIOPEP and computer-aided matching tools can rapidly screen candidate bioactive peptides, whose actual biological activity is further verified through in vitro experiments to explore novel ACE inhibitory components [82-83]. Furthermore, systematic assessment and predictive analysis of the physicochemical properties, primary structures, toxicological risks and allergenicity of most ACE inhibitory peptides can be performed, so as to comprehensively guarantee the safety of final functional food products [84]. The combined application of these advanced separation and identification technologies greatly promotes the basic research and industrial development of rice-derived hypotensive functional foods and other bioactive food products.
四. Research Prospects and Limitations of Rice-Derived ACEIPs
The production of ACEIPs from rice and its processing by-products via enzymatic hydrolysis, microbial fermentation or combined biotechnological strategies presents enormous development potential. Consumers have gradually recognized the unique functional advantages and nutritional value of rice-derived functional products. Nevertheless, compared with animal-derived and other plant-derived bioactive peptides, research on rice-based ACEIPs prepared by enzymatic hydrolysis and fermentation remains relatively limited, leaving substantial room for in-depth exploration and industrial application.
The structure-activity relationship of ACEIPs and their functional regulatory mechanisms require further systematic investigation. In particular, in vivo metabolic pathways and clinical evidence of fermented rice-derived ACEIPs are still insufficient. Future research on the bioactivity of ACEIPs is expected to rely on multi-omics technologies including genomics and metabolomics to clarify their biosynthesis mechanisms and structural contributions to physiological functions. Transcriptomics and other molecular biological approaches, combined with computational simulation software, can be utilized to analyze the interactive relationships among fermentative microorganisms, optimize the targeted biosynthesis of rice-derived bioactive peptides, and elucidate their intracellular metabolism and transmembrane transport mechanisms.
Further in-depth research is urgently required to enhance peptide production efficiency and oral bioavailability, evaluate the in vivo bioaccessibility and long-term hypotensive efficacy of rice-derived ACEIPs. Collectively, ongoing mechanistic and applied research will provide solid theoretical and technical support for the innovative development and clinical auxiliary application of health care products and adjuvant antihypertensive agents based on rice bioactive peptides.
References:
[1] DZAU V J, HODGKINSON C P. Precision hypertension[J]. Hypertension, 2024, 81(4): 702-708.
[2] OLFAT A, MOSTAGHIM T, SHAHRIARI S, et al. Extraction of bioactive compounds of Hypnea flagelliformis by ultrasound-assisted extraction coupled with natural deep eutectic solvent and enzyme inhibitory activity[J]. Algal Research, 2024, 78: 103388.
[3] OPARIL S, ACELAJADO M C, BAKRIS G L, et al. Hypertension[J]. Nature Reviews Disease Primers, 2018, 4: 18014.
[4] WANG J W, YE Y Y, CHEN X F, et al. Diverse trends in antihypertensive medication usage among U.S. adults with hypertension by socioeconomic status and comorbidities, 1999—2020[J]. Blood Pressure, 2025, 34(1): 2506081.
[5] FABIAN I M, MADDOX K, ROBICHEAUX C, et al. Stevens-Johnson syndrome from combined allopurinol and angiotensin-converting enzyme inhibitors: A narrative review[J]. Cureus, 2024, 16(1): e51899.
[6] VILLANUEVA A, RIVERO-PINO F, MARTIN M E, et al. Identification of the bioavailable peptidome of chia protein hydrolysate and the in silico evaluation of its antioxidant and ACE inhibitory potential[J]. Journal of Agricultural and Food Chemistry, 2024, 72(6): 3189-3199.
[7] RAMAKRISHNAN V V, HOSSAIN A, DAVE D, et al. Salmon processing discards: a potential source of bioactive peptides-a review[J]. Food Production, Processing and Nutrition, 2024, 6(1): 22.
[8] XIANG L, QIU Z C, ZHAO R J, et al. Advancement and prospects of production, transport, functional activity and structure-activity relationship of food-derived angiotensin converting enzyme (ACE) inhibitory peptides[J]. Critical Reviews in Food Science and Nutrition, 2023, 63(10): 1437-1463.
[9] HERES A, YOKOYAMA I, GALLEGO M, et al. Antihypertensive potential of sweet Ala-Ala dipeptide and its quantitation in dry-cured ham at different processing conditions[J]. Journal of Functional Foods, 2021, 87: 104818.
[10] SHANMUGAM V P, KAPILA S, KEMGANG T S, et al. Isolation and characterization of angiotensin converting enzyme inhibitory peptide from buffalo casein[J]. International Journal of Peptide Research and Therapeutics, 2021, 27(2): 1481-1491.
[11] KAISER S, MARTIN M, LUNOW D, et al. Tryptophan-containing dipeptides are bioavailable and inhibit plasma human angiotensin-converting enzyme in vivo[J]. International Dairy Journal, 2016, 52: 107-114.
[12] ZHANG D J, JIANG K, LUO H, et al. Replacing animal proteins with plant proteins: Is this a way to improve quality and functional properties of hybrid cheeses and cheese analogs?[J]. Comprehensive Reviews in Food Science and Food Safety, 2024, 23(1): e13262.
[13] PREKUMAR J, MALINI M, JOSHY A. A critical review on food protein-derived antihypertensive peptides[J]. Drug Invention Today, 2019, 12(3): 474-479.
[14] CHUKWUMA C I. Antioxidative, metabolic and vascular medicinal potentials of natural products in the non-edible wastes of fruits belonging to the Citrus and Prunus genera: A review[J]. Plants, 2024, 13(2): 191.
[15] FARIA-COSTA G, LEITE-MOREIRA A, HENRIQUES-COELHO T. Efeitos cardiovasculares do receptor tipo 2 da angiotensina[J]. Revista Portuguesa de Cardiologia, 2014, 33(7/8): 439-449.
[16] METLOCK F E, HINNEH T, BENJASIRISAN C, et al. Impact of social determinants of health on hypertension outcomes: A systematic review[J]. Hypertension, 2024, 81(8): 1675-1700.
[17] HALLBERG M, LARHED M. From angiotensin IV to small peptidemimetics inhibiting insulin-regulated aminopeptidase[J]. Frontiers in Pharmacology, 2020, 11: 590855.
[18] SUN S Q, XU X T, SUN X, et al. Preparation and identification of ACE inhibitory peptides from the marine macroalga Ulva intestinalis[J]. Marine Drugs, 2019, 17(3): 179.
[19] TOOPCHAM T, MES J J, WICHERS H J, et al. Bioavailability of angiotensin I-converting enzyme (ACE) inhibitory peptides derived from Virgibacillus halodenitrificans SK1-3-7 proteinases hydrolyzed tilapia muscle proteins[J]. Food Chemistry, 2017, 220: 190-197.
[20] MIRALLES B, AMIGO L, RECIO I. Critical review and perspectives on food-derived antihypertensive peptides[J]. Journal of Agricultural and Food Chemistry, 2018, 66(36): 9384-9390.
[21] JAO C L, HUANG S L, HSU K C. Angiotensin I-converting enzyme inhibitory peptides: Inhibition mode, bioavailability, and antihypertensive effects[J]. BioMedicine, 2012, 2(4): 130-136.
[22] CIAU-SOLÍS N, RODRÍGUEZ-CANTO W, FERNÁNDEZ-MARTÍNEZ L, et al. Inhibitory activity of angiotensin-I converting enzyme (ACE-I) from partially purified Phaseolus lunatus peptide fractions[J]. Process Biochemistry, 2024, 139: 44-50.
[23] TAGA Y, HAYASHIDA O, ASHOUR A, et al. Characterization of angiotensin-converting enzyme inhibitory activity of X-hyp-gly-type tripeptides: Importance of collagen-specific prolyl hydroxylation[J]. Journal of Agricultural and Food Chemistry, 2018, 66(33): 8737-8743.
[24] LIU C, ZHANG Z J, SHANG Y T, et al. Progress in the preparation, identification and biological activity of walnut peptides[J]. Journal of Future Foods, 2024, 4(3): 205-220.
[25] XIANG L, ZHENG Z J, GUO X J, et al. Two novel angiotensin I-converting enzyme inhibitory peptides from garlic protein: In silico screening, stability, antihypertensive effects in vivo and underlying mechanisms[J]. Food Chemistry, 2024, 435: 137537.
[26] WANG X M, CHEN H X, FU X G, et al. A novel antioxidant and ACE inhibitory peptide from rice bran protein: Biochemical characterization and molecular docking study[J]. LWT-Food Science and Technology, 2017, 75: 93-99.
[27] LEE S Y, HUR S J. Antihypertensive peptides from animal products, marine organisms, and plants[J]. Food Chemistry, 2017, 228: 506-517.
[28] Chen S R, Wei T, Li Q, et al. Process optimization and dynamic analysis of ACE inhibitory peptides produced by fermenting buffalo milk with Lactobacillus plantarum Strain B[J]. Cereals & Oils, 2024, 37(12): 89-96.
[29] LI H J, WANG Y S, WANG Y N, et al. Mechanical study of alisol B23-acetate on methionine and choline deficient diet-induced nonalcoholic steatohepatitis based on untargeted metabolomics[J]. Biomedical Chromatography, 2024, 38(1): e5763.
[30] BUTTERWORTH R F, CANBAY A. Hepatoprotection by L-ornithine L-aspartate in non-alcoholic fatty liver disease[J]. Digestive Diseases, 2019, 37(1): 63-68.
[31] MONDAL S, DAS S, NANDI A K. A review on recent advances in polymer and peptide hydrogels[J]. Soft Matter, 2020, 16(6): 1404-1454.
[32] FENG L, WANG Y, YANG J, et al. Overview of the preparation method, structure and function, and application of natural peptides and polypeptides[J]. Biomedecine & Pharmacotherapie, 2022, 153: 113493.
[33] TYAGI A, CHELLIAH R, BANAN-MWINE DALIRI E, et al. Antioxidant activities of novel peptides from Limosilactobacillus reuteri fermented brown rice: A combined in vitro and in silico study[J]. Food Chemistry, 2023, 404(Pt B): 134747.
[34] DALIRI E B, LEE B H, OH D H. Current trends and perspectives of bioactive peptides[J]. Critical Reviews in Food Science and Nutrition, 2018, 58(13): 2273-2284.
[35] KONTANI N, OMAE R, KAGEBAYASHI T, et al. Characterization of Ile-His-Arg-Phe, a novel rice-derived vasorelaxing peptide with hypotensive and anorexigenic activities[J]. Molecular Nutrition & Food Research, 2014, 58(2): 359-364.
[36] KUMAR A, SINGH N, JOSHI R. Evaluation of antioxidant and ACE inhibitory peptides in rice bran protein hydrolysate using non-targeted UHPLC-QTOF-IMS profiling, MALDI-TOF-TOF-MS/MS, and molecular docking study[J]. International Journal of Food Science & Technology, 2024, 59(6): 4300-4314.
[37] KUBA M, TANAKA K, SESOKO M, et al. Angiotensin I-converting enzyme inhibitory peptides in red-mold rice made by Monascus purpureus[J]. Process Biochemistry, 2009, 44(10): 1139-1143.
[38] KANG M G, KIM J H, AHN B H, et al. Characterization of new antihypertensive angiotensin I-converting enzyme inhibitory peptides from Korean traditional rice wine[J]. Journal of Microbiology and Biotechnology, 2012, 22(3): 339-342.
[39] NEMOTO Y, MATSUO Y, SHIODA K, et al. Compositional differences, antioxidant activity, angiotensin-converting enzyme inhibition, and sensory characteristics of kamaboko fermented in wheat, rice, brown rice, and soybean koji[J]. Fisheries Science, 2020, 86(3): 581-593.
[40] HU Y, SUN-WATERHOUSE D, LIU P Z, et al. Modification of rice protein with glutaminase for improved structural and sensory properties[J]. International Journal of Food Science and Technology, 2019, 54(7): 2458-2467.
[41] DONG J J, WANG S, YIN X Y, et al. Angiotensin I converting enzyme (ACE) inhibitory activity and antihypertensive effects of rice peptides[J]. Food Science and Human Wellness, 2022, 11(6): 1539-1543.
[42] DING Y H, HAN P, MA H L, et al. Process control of rice protein enzymolysis by field monitoring[J]. Current Research in Biotechnology, 2024, 7: 100171.
[43] LONG Y H, GAO Z, QIU C H, et al. Rice dreg protein as a high-quality plant-based protein resource: Characterization of structural, functional, and in vitro digestive properties[J]. Cereal Chemistry, 2024, 101(4): 785-797.
[44] SUWANNACHOT P, THAWORNCHINSOMBUT S, JONGJAREONRAK A, et al. Supplementation with rice bran hydrolysates reduces oxidative stress and improves lipid profiles in adult dogs[J]. Journal of Veterinary Medical Science, 2023, 85(7): 727-734.
[45] JUNSARA K, YUPANQUI C T, KAWEE-AI A, et al. Fortification of crude protein extract from Sung Yod and Hom rajinee rice brans in the development of functional jelly products[J]. Foods, 2023, 12(6): 1138.
[46] KUKONGVIRIVAPAN U, JAN-ON G, TUBSAKUL A, et al. Sang-Yod rice bran hydrolysates alleviate hypertension, endothelial dysfunction, vascular remodeling, and oxidative stress in nitric oxide deficient hypertensive rats[J]. Asian Pacific Journal of Tropical Biomedicine, 2021, 11(1): 10-19.
[47] ZHANG L Y, MIAO J Y, GUO J B, et al. Two novel angiotensin I-converting enzyme (ACE) inhibitory peptides from rice (Oryza sativa L.) bran protein[J]. Journal of Agricultural and Food Chemistry, 2023, 71(9): 4153-4162.
[48] WU Y, ZHANG X X, CUI H W, et al. Isolation, identification and activity analysis of antioxidant peptides from rice wine lees[J]. Journal of Food Measurement and Characterization, 2023, 18(2): 1528-1542.
[49] SU H X, FAN W L, XU Y, et al. Preparation, identification, and molecular docking of novel angiotensin-converting enzyme inhibitory peptides derived from rice-based distillers’ spent cakes[J]. Journal of the Science of Food and Agriculture, 2024, 104(11): 6506-6517.
[50] WONGSA P, YUENYONGRATTANAKORN K, PONGVACHIRINT W, et al. Improving anti-hypertensive properties of plant-based alternatives to yogurt fortified with rice protein hydrolysate[J]. Heliyon, 2022, 8(10): e11087.
[51] NGAMSUK S, HSU J L, HUANG T C. Ultrasonication of milky stage rice milk with bioactive peptides from rice bran: Its bioactivities and absorption[J]. Food and Bioprocess Technology, 2020, 13(3): 462-474.
[52] DOU B X, LIU Y, LIU Y M. Isolation and characterization of angiotensin I converting enzyme (ACE) inhibitory peptides from rice bran proteins and evaluation of activity and stability[J]. Pakistan Journal of Zoology, 2020, 52(4): 1383-1391.
[53] SHOBAKO N, OGAWA Y, ISHIKADO A, et al. A novel antihypertensive peptide identified in thermolysin-digested rice bran[J]. Molecular Nutrition & Food Research, 2018, 62(4): 1700732.
[54] SHOBAKO N, ISHIKADO A, OGAWA Y, et al. Vasorelaxant and antihypertensive effects that are dependent on the endothelial NO system exhibited by rice bran-derived tripeptide[J]. Journal of Agricultural and Food Chemistry, 2019, 67(5): 1437-1442.
[55] SUWANNAPAN O, WACHIRATTANAPONGMETEE K, THAWORNCHINSOMBUT S, et al. Angiotensin-I-converting enzyme (ACE)-inhibitory peptides from Thai jasmine rice bran protein hydrolysates[J]. International Journal of Food Science & Technology, 2020, 55(6): 2441-2450.
[56] SARRINGKARIN W, LAOKULDILOK T. Optimization of the production conditions of glutinous rice bran protein hydrolysate with antioxidative properties[J]. Chiang Mai University Journal of Natural Sciences, 2017, 16(1): 0001.
[57] PIOTROWICZ I B B, GARCÉS-RIMÓN M, MORENO-FERNÁNDEZ S, et al. Antioxidant, angiotensin-converting enzyme inhibitory properties and blood-pressure-lowering effect of rice bran protein hydrolysates[J]. Foods, 2020, 9(6): 812.
[58] SELAMASSAKUL O, LAOHAKUNJIT N, KERDCHOEUEN O, et al. Bioactive peptides from brown rice protein hydrolyzed by bromelain: Relationship between biofunctional activities and flavor characteristics[J]. Journal of Food Science, 2020, 85(3): 707-717.
[59] CÁCERES P J, PEÑAS E, MARTÍNEZ-VILLALUENGA C, et al. Development of a multifunctional yogurt-like product from germinated brown rice[J]. LWT-Food Science and Technology, 2019, 99: 306-312.
[60] AHMADIFARD N, MURUETA J H C, ABEDIAN-KENARI A, et al. Comparison the effect of three commercial enzymes for enzymatic hydrolysis of two substrates (rice bran protein concentrate and soybean protein) with SDS-PAGE[J]. Journal of Food Science and Technology, 2016, 53(2): 1279-1284.
[61] CHEN L, WANG L L, LI J K, et al. Antihypertensive potential of fermented milk: The contribution of lactic acid bacteria proteolysis system and the resultant angiotensin-converting enzyme inhibitory peptide[J]. Food & Function, 2021, 12(22): 11121-11131.
[62] JUILLARD V, LOPEZ-KLEINE L, MONNET V. Lactic acid bacteria: Proteolytic systems[M]//MCSWEENEY P L H, MCNAMARA J P. Encyclopedia of Dairy Sciences (3rd ed.). Oxford: Academic Press, 2022: 249-255.
[63] MUSTAFA M H, SOLEIMANIAN-ZAD S, SHEIKHZEINODDIN M. Characterization of a trypsin-like protease 1 produced by a probiotic Lactobacillus plantarum subsp. plantarum PTCC 1896 from skimmed milk based medium[J]. LWT-Food Science and Technology, 2020, 119: 108818.
[64] SOLIERI L, RUTELLA G S, TAGLIAZUCCHI D. Impact of non-starter lactobacilli on release of peptides with angiotensin-converting enzyme inhibitory and antioxidant activities during bovine milk fermentation[J]. Food Microbiology, 2015, 51: 108-116.
[65] YIN L B, LIU Y L, HE P, et al. Optimization of antihypertensive peptide preparation by Lactobacillus plantarum fermentation of soy whey[J]. Journal of Biobased Materials and Bioenergy, 2021, 15(2): 238-243.
[66] SINGH B P, VIJ S. Growth and bioactive peptides production potential of Lactobacillus plantarum Strain C2 in soy milk: A LC-MS/MS based revelation for peptides biofunctionality[J]. LWT-Food Science and Technology, 2017, 86: 293-301.
[67] ZAREIAN M, OSKOUEIAN E, FORGHANI B, et al. Production of a wheat-based fermented rice enriched with γ-aminobutyric acid using Lactobacillus plantarum MNZ and its antihypertensive effects in spontaneously hypertensive rats[J]. Journal of Functional Foods, 2015, 16: 194-203.
[68] PUSPITOJATI E, CAHYANTO M N, MARSONO Y, et al. Changes in amino acid composition during fermentation and its effects on the inhibitory activity of angiotensin-I converting enzyme of jack bean tempe following in vitro gastrointestinal digestion[J]. Journal of Food and Nutrition Research, 2019, 58(4): 319-327.
[69] GE H J, ZHANG Z K, XIAO J X, et al. Release of Leu-Pro-Pro from corn gluten meal by fermentation with a Lactobacillus helveticus strain[J]. Journal of the Science of Food and Agriculture, 2022, 102(3): 1095-1104.
[70] SEKINE T, NAGAI H, HAMADA-SATO N. Antihypertensive and probiotic effects of hidakakombu (Saccharina angustata) fermented by Lacticaseibacillus casei 001[J]. Foods, 2021, 10(9): 2048.
[71] PARMAR H, HATI S, SAKURE A. In vitro and in silico analysis of novel ACE-inhibitory bioactive peptides derived from fermented goat milk[J]. International Journal of Peptide Research and Therapeutics, 2018, 24(3): 441-453.
[72] WU Q, LI Y, PENG K, et al. Isolation and characterization of three antihypertension peptides from the mycelia of Ganoderma lucidum (Agaricomycetes)[J]. Journal of Agricultural and Food Chemistry, 2019, 67(29): 8149-8159.
[73] LU F, ALENYOREGE E A, OUYANG N N, et al. Simulated natural and high temperature solid-state fermentation of soybean meal: A comparative study regarding microorganisms, functional properties and structural characteristics[J]. LWT-Food Science and Technology, 2022, 159: 113125.
[74] MOAYEDI A, MORA L, ARISTOY M C, et al. ACE-inhibitory and antioxidant activities of peptide fragments obtained from tomato processing by-products fermented using Bacillus subtilis: Effect of amino acid composition and peptides molecular mass distribution[J]. Applied Biochemistry and Biotechnology, 2016, 181(1): 48-64.
[75] TAMAM B, SYAH D, SUHARTONO M T, et al. Proteomic study of bioactive peptides from tempe[J]. Journal of Bioscience and Bioengineering, 2019, 128(2): 241-248.
[76] ARDIANSYAH A. A short review: Bioactivity of fermented rice bran[J]. Journal of Oleo Science, 2021, 70(11): 1565-1574.
[77] HU J F, WANG H Y, WENG N H, et al. Novel angiotensin-converting enzyme and pancreatic lipase oligopeptide inhibitors from fermented rice bran[J]. Frontiers in Nutrition, 2022, 9: 1010005.
[78] ALAUDDIN M, SHIRAKAWA H, KOSEKI T, et al. Fermented rice bran supplementation mitigates metabolic syndrome in stroke-prone spontaneously hypertensive rats[J]. BMC Complementary and Alternative Medicine, 2016, 16(1): 442.
[79] GUO J B, LU A C, SUN Y N, et al. Purification and identification of antioxidant and angiotensin converting enzyme-inhibitory peptides from Guangdong glutinous rice wine[J]. LWT-Food Science and Technology, 2022, 169: 113953.
[80] MISHRA B K, DAS S, PRAJAPATI J B, et al. Bio-functional properties and storage study of ‘Chubitchi’- a fermented rice beverage of Garo Hills, Meghalaya[J]. Indian Journal of Traditional Knowledge, 2021, 20(2): 498-511.
[81] DALIRI E B, OFOSU F K, CHELLIAH R, et al. Untargeted metabolomics of fermented rice using UHPLC QTOF MS/MS reveals an abundance of potential antihypertensive compounds[J]. Foods, 2020, 9(8): 1007.
[82] POOJA K, RANI S, PRAKASH B. In silico approaches towards the exploration of rice bran proteins-derived angiotensin-I-converting enzyme inhibitory peptides[J]. International Journal of Food Properties, 2017, 20(2): 2178-2191.
[83] Xia X Y, Wen C X, Cao W H, et al. Screening of ACE inhibitory peptides from enzymatic hydrolysates of Pinctada martensii meat based on surface plasmon resonance technology[J]. Food Science, 2025, 46(7): 143-150.
[84] WU N, LI P, SHUANG Q, et al. Screening and molecular dynamics simulation of ACE inhibitory tripeptides derived from milk fermented with Lactobacillus delbrueckii QS306[J]. Food & Function, 2024, 15(5): 2655-2667.
Post time: Apr-28-2026